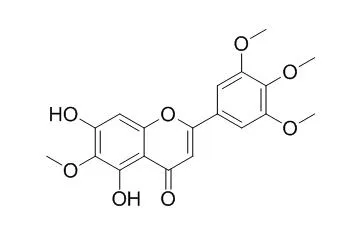
| Structure Identification: |
| Biomed Res Int. 2017;2017:7375615. | | Inhibitory Effect of Chemical Constituents Isolated from Artemisia iwayomogi on Polyol Pathway and Simultaneous Quantification of Major Bioactive Compounds.[Pubmed: 28512639 ] | Blocking the polyol pathway plays an important role preventing diabetic complications. Therefore, aldose reductase (AR) and advanced glycation endproducts (AGEs) formation has significant effect on diabetic complications. Artemisia iwayomogi has long been used as treatment of various diseases in Korea. However, no literatures have reported on AR and AGEs formation inhibitory activities of A. iwayomogi. For these reasons, we aimed to assess that A. iwayomogi had potential as anti-diabetic complications agents.
METHODS AND RESULTS:
We led to isolation of two coumarins (1 and 2), nine flavonoids (3-11), five caffeoylquinic acids (12-16), three diterpene glycosides (17-19), and one phenolic compound (20) from A. iwayomogi. Among them, hispidulin (4), 6-methoxytricin (6), Arteanoflavone (7), quercetin-3-gentiobioside (10), 1,3-di-O-caffeoylquinic acid (13), and suavioside A (18) were first reported on the isolation from A. iwayomogi. Not only two coumarins (1 and 2), nine flavonoids (3-11), and five caffeoylquinic acids (12-16) but also extracts showed significant inhibitor on AR and AGEs formation activities. We analyzed contents of major bioactive compounds in Korea's various regions of A. iwayomogi.
CONCLUSIONS:
Overall, we selected Yangyang, Gangwon-do, from June, which contained the highest amounts of bioactive compounds, as suitable areas for cultivating A. iwayomogi as preventive or therapeutic agent in the treatment of diabetic complications. | | Phytochemistry, 1988, 27(10):3155-3159. | | Flavonoids and other phenolics from Artemisia hispanica.[Reference: WebLink] | | Extraction of aerial parts of Artemisia hispanica and chromatographic separation yielded chrysosplenetin, chrysosplenol D, 5,7,3′,4′-tetrahydroxy-6,5′-dimethoxyflavone, 5,7,3′-trihydroxy-6,4′,5′-trimethoxyflavone, Arteanoflavone, cirsilineol, penduletin, axillarin, jaceosidin, apigenin, luteolin, p-hydroxyacetophenone, 4-(p-hydroxyphenyl) butan-2-one, methyl caffeate, esculetin, apigenin 7-glucoside, luteolin 7-glucoside and the new flavonoids 5,3′,4′-trihydroxy-6,7,5′-trimethoxyflavone and isoetin 5′-glucoside. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)