| In vitro: |
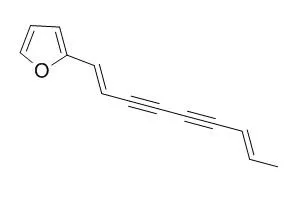
| Chem Biodivers. 2015 Apr;12(4):593-8. | | Insecticidal and Repellant Activities of Polyacetylenes and Lactones Derived from Atractylodes lancea Rhizomes.[Pubmed: 25879503] | During a screening program for new agrochemicals from Chinese medicinal herbs and local wild plants, the petroleum ether (PE) extract of Atractylodes lancea (Thunb.) rhizomes was found to possess repellent and contact activities against Tribolium castaneum adults.
METHODS AND RESULTS:
Bioactivity-directed chromatographic separation of PE extract on repeated silica-gel columns led to the isolation of two polyacetylenes, Atractylodin and Atractylodinol (1 and 2, resp.), and two lactones, atractylenolides II and III (3 and 4, resp.). The structures of the compounds were elucidated based on NMR spectra. The four isolated compounds were evaluated for their insecticidal and repellent activities against T. castaneum. Atractylodin exhibited strong contact activity against T. castaneum adults with a LD50 value of 1.83 μg/adult. Atractylodin and atractylenolide II also possessed strong repellenct activities against T. castaneum adults. After 4-h exposure, >90% repellency was achieved with Atractylodin at a low concentration of 0.63 μg/cm(2) .
CONCLUSIONS:
The results indicated that Atractylodin (1) and atractylenolide II (3) have a good potential as a source for natural repellents, and 1 has the potential to be developed as natural insecticide. | | Chinese Traditional & Herbal Drugs, 2014, 45(5): 701-8. | | Signal transduction of atractylodin biosynthesis in Atractylodes lancea cell induced by endophytic fungal elicitor mediated with nitric oxide followed by salicylic acid[Reference: WebLink] | To investigate the signal molecules and signal transduction involved in endophytic fungal elicitor-induced Atractylodin biosynthesis and the effect of an endophytic fungal elicitor on the key enzyme activity in Atractylodes lancea.
METHODS AND RESULTS:
Content changes of nitric oxide (NO), salicylic acid (SA), and Atractylodin were detected under the endophytic fungal elicitor treatment by plant cell suspension culture technology. Results The endophytic fungal elicitor remarkably promoted NO burst and the biosynthesis of SA and atractyodin by activating nitric oxide synthase (NOS), phenylalanine ammonia lyase (PAL), and acetyl coenzyme A carboxylase (ACC), respectively. NOS inhibitor PBITU could inhibit the NO and SA accumulation and the atractyodin biosynthesis induced by the elicitor. And atractyodin biosynthesis could also be triggered by exogenous NO or SA. The results indicated that NO and SA were the necessary signal molecules and NO burst was mediated by NOS induced by endophytic fungal elicitor. NO quencher cPITO could effectively remove NO burst in A. lancea cell induced by endophytic fungal elicitor and notably inhibit the biosynthesis promotion of SA and atractyodin in A. lancea cell induced by endophytic fungal elicitor. Exogenous SNP could reverse the cPITO inhibition on the activity of PAL and ACC and the synthesis of SA and Atractylodin. This suggested that NO was an upstream signal molecule mediated endophytic fungal elicitor to accelerate the biosynthesis of SA and atractyodin.
CONCLUSIONS:
Endophytic fungal elicitor mediated through NO followed by SA could promote atractyodin biosynthesis by activating ACC in A. Lancea.
|
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)