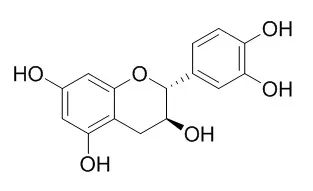
| Description: |
Catechin, a cyclooxygenase-1 (COX-1) inhibitor with an IC50 of 1.4 μM, which has antiangiogenic, antitumor, antioxidant, UV-protective, anti-aging, phytotoxic, antimicrobial, and antiviral effects. Catechin shows its potential as biobased active packaging for fatty food, and exerts cardioprotection through treating many kinds of angiocardiopathy. |
| In vitro: |
| Pharm Dev Technol. 2014 Jun;19(4):395-400. | | Effect of emulsification on the skin permeation and UV protection of catechin.[Pubmed: 23639253] | An anti-aging effect may be obtained by skin application of tea Catechins (Camellia sinensis) since they have high ultraviolet (UV)-protection activity.
METHODS AND RESULTS:
In this study, the skin permeation of Catechin (C), epiCatechin (EC), epigalloCatechin (EGC), epiCatechin gallate (ECg) and epigalloCatechin gallate (EGCg) was determined and compared, and the effect of emulsification on the skin permeation of C was measured. The UV-protective effect of C was also determined. The in vitro skin permeability of each Catechin derivative was determined using side-by-side diffusion of cells. The UV-protective effect of C was determined by applying different concentrations of C to the solution or emulsion on a three-dimensional cultured human skin model or normal human epidermal keratinocytes with UV-irradiation. ECg and EGCg with gallate groups showed lower skin permeability than C, EC and EGC without gallate groups, suggesting that the skin permeability of Catechin derivatives may be dependent on the existence of a gallate group. Interestingly, the skin permeation of C was increased by an o/w emulsification. In addition, the C emulsion showed a significantly higher UV-protective effect by C than that with its aqueous solution.
CONCLUSIONS:
These results suggest that the o/w emulsion of Catechin derivatives is probably useful as a cosmetic formulation with anti-aging efficacy. | | Biochem Pharmacol. 2011 Dec 15;82(12):1807-21. | | Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications.[Pubmed: 21827739 ] | An expanding body of preclinical evidence suggests EGCG, the major Catechin found in green tea (Camellia sinensis), has the potential to impact a variety of human diseases.
METHODS AND RESULTS:
Apparently, EGCG functions as a powerful antioxidant, preventing oxidative damage in healthy cells, but also as an antiangiogenic and antitumor agent and as a modulator of tumor cell response to chemotherapy. Much of the cancer chemopreventive properties of green tea are mediated by EGCG that induces apoptosis and promotes cell growth arrest by altering the expression of cell cycle regulatory proteins, activating killer caspases, and suppressing oncogenic transcription factors and pluripotency maintain factors. In vitro studies have demonstrated that EGCG blocks carcinogenesis by affecting a wide array of signal transduction pathways including JAK/STAT, MAPK, PI3K/AKT, Wnt and Notch. EGCG stimulates telomere fragmentation through inhibiting telomerase activity. Various clinical studies have revealed that treatment by EGCG inhibits tumor incidence and multiplicity in different organ sites such as liver, stomach, skin, lung, mammary gland and colon. Recent work demonstrated that EGCG reduced DNMTs, proteases, and DHFR activities, which would affect transcription of TSGs and protein synthesis. EGCG has great potential in cancer prevention because of its safety, low cost and bioavailability. In this review, we discuss its cancer preventive properties and its mechanism of action at numerous points regulating cancer cell growth, survival, angiogenesis and metastasis.
CONCLUSIONS:
Therefore, non-toxic natural agent could be useful either alone or in combination with conventional therapeutics for the prevention of tumor progression and/or treatment of human malignancies. | | Cytotechnology . 2018 Feb;70(1):245-259. | | Catechin ameliorates doxorubicin-induced neuronal cytotoxicity in in vitro and episodic memory deficit in in vivo in Wistar rats[Pubmed: 28900743] | | Cognitive dysfunction by chemotherapy compromises the quality of life in cancer patients. Tea polyphenols are known chemopreventive agents. The present study was designed to evaluate the neuroprotective potential of (+) Catechin hydrate (Catechin), a tea polyphenol, in IMR-32 neuroblastoma cells in vitro and alleviation of episodic memory deficit in Wistar rats in vivo against a widely used chemotherapeutic agent, Doxorubicin (DOX). In vitro, neuroprotective studies were assessed in undifferentiated IMR-32 cells using percentage viability and in differentiated cells by neurite length. These studies showed Catechin increased percentage viability of undifferentiated IMR-32 cells. Catechin pretreatment also showed an increase in neurite length of differentiated cells. In vivo neuroprotection of Catechin was evaluated using novel object recognition task in time-induced memory deficit model at 50, 100 and 200 mg/kg dose and DOX-induced memory deficit models at 100 mg/kg dose. The latter model was developed by injection of DOX (2.5 mg/kg, i.p.) in 10 cycles over 50 days in Wistar rats. Catechin showed a significant reversal of time-induced memory deficit in a dose-dependent manner and prevention of DOX-induced memory deficit at 100 mg/kg. In addition, Catechin treatment showed a significant decrease in oxidative stress, acetylcholine esterase and neuroinflammation in the hippocampus and cerebral cortex in DOX-induced toxicity model. Hence, Catechin may be a potential adjuvant therapy for the amelioration of DOX-induced cognitive impairment which may improve the quality of life of cancer survivors. This improvement might be due to the elevation of antioxidant defense, prevention of neuroinflammation and inhibition of acetylcholine esterase enzyme. |
|
| In vivo: |
| Am J Clin Nutr. 2001 Aug;74(2):227-32. | | Catechin intake might explain the inverse relation between tea consumption and ischemic heart disease: the Zutphen Elderly Study.[Pubmed: 11470725] | Epidemiologic studies suggest that tea consumption may reduce the risk of cardiovascular diseases, but results are inconsistent. Catechins, which belong to the flavonoid family, are the main components of tea and may be responsible for the alleged protective effect. Taking Catechin sources other than tea into account might clarify the reported associations.
METHODS AND RESULTS:
The objective was to evaluate the association between Catechin intake and the incidence of and mortality from ischemic heart disease and stroke.
We evaluated the effect of a high Catechin intake by using data from the Zutphen Elderly Study, a prospective cohort study of 806 men aged 65-84 y at baseline in 1985.
The mean (+/-SD) Catechin intake at baseline was 72 +/- 47.8 mg, mainly from black tea, apples, and chocolate. A total of 90 deaths from ischemic heart disease were documented. Catechin intake was inversely associated with ischemic heart disease mortality; the multivariate-adjusted risk ratio in the highest tertile of intake was 0.49 (95% CI: 0.27, 0.88; P for trend: 0.017). After multivariate adjustment, Catechin intake was not associated with the incidence of myocardial infarction (risk ratio in the highest tertile of intake: 0.70; 95% CI: 0.39, 1.26; P for trend: 0.232). After adjustment for tea consumption and flavonol intake, a 7.5-mg increase in Catechin intake from sources other than tea was associated with a tendency for a 20% reduction in ischemic heart disease mortality risk (P = 0.114). There was no association between Catechin intake and stroke incidence or mortality.
CONCLUSIONS:
Catechins, whether from tea or other sources, may reduce the risk of ischemic heart disease mortality but not of stroke. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)