| In vitro: |
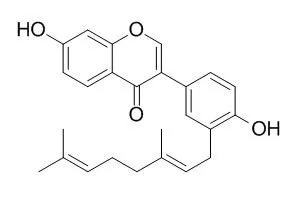
| Carbohydr Res. 2017 Jun 29;446-447:61-67. | | Enzymatic synthesis of novel corylifol A glucosides via a UDP-glycosyltransferase.[Pubmed: 28528234 ] | Corylifol A, a member of the isoflavone subclass of isoflavonoids, has long been considered to have various biological activities.
METHODS AND RESULTS:
Here, we sought to synthesize Corylifol A glucosides by the in vitro glucosylation reaction using the UDP-glycosyltransferase YjiC from Bacillus licheniformis DSM 13, and obtained two novel glucosides: Corylifol A-4',7-di-O-beta-d-glucopyranoside (1) and Corylifol A-4'-O-beta-d-glucopyranoside (2). To improve the yield of the products, the reaction time, concentration of UDP-glucose, and pH of the buffer were optimized. The Michaelis constant (Km) was calculated to be 2.88 mM, and the maximal velocity (Vmax) was calculated to be 77.32 nmol/min/mg for UDP-glycosyltransferase. Meanwhile, the water-solubility of compounds 1 and 2 was approximately 27.03 and 15.13 times higher, respectively, than that of their parent compound Corylifol A. Additionally, the Corylifol A glycosylated products exhibited the highest stability at pH 9.6 and better temperature stability than Corylifol A at 40, 60, 80 and 100 °C. In addition, cytotoxicity activity assays against three human tumor cell lines, only Corylifol A showed moderate anti-proliferative activity.
CONCLUSIONS:
Overall, this work demonstrates that glycosylation can enhance the water solubility and stability of promising compounds, with potential for further development and application. | | J Asian Nat Prod Res. 2013;15(6):624-30. | | Cytotoxic constituents from Psoralea corylifolia.[Pubmed: 23659434 ] | Bioassay directed isolation of the EtOAc extract from a traditional Chinese medicine Psoralea corylifolia resulted in the purification of two isoflavonoids, corylifols D (1) and E (2), along with four known ones.
METHODS AND RESULTS:
The structures of 1 and 2 were determined by extensive 1D and 2D NMR and MS data analyses. When tested against HepG2 and Hep3B hepatocellular carcinoma cell lines, Corylifol A (4) displayed IC50 values of 4.6 and 13.5 μg/ml, respectively. | | Planta Med. 2012 Jun;78(9):903-6. | | Phenolic compounds isolated from Psoralea corylifolia inhibit IL-6-induced STAT3 activation.[Pubmed: 22573369 ] | Inhibiting interleukin-6 (IL-6) has been postulated as an effective therapy in the pathogenesis of several inflammatory diseases.
METHODS AND RESULTS:
In this study, seven flavonoids were isolated from the methanol extracts of Psoralea corylifolia by bioactivity-guided fractionation. The structures of bakuchiol (1), bavachinin (2), neobavaisoflavone (3), Corylifol A (4), corylin (5), isobavachalcon (6), and bavachin (7) were determined by spectroscopic analysis (1H-, 13C- NMR and MS). We demonstrated that compounds 1-7 showed an inhibitory effect on IL-6-induced STAT3 promoter activity in Hep3B cells with IC50 values of 4.57 ± 0.45, 3.02 ± 0.53, 2.77 ± 0.02, 0.81 ± 0.15, 1.37 ± 0.45, 2.45 ± 0.13, and 4.89 ± 0.05 μΜ, respectively. These compounds also inhibited STAT3 phosphorylation induced by IL-6 in Hep3B cells.
CONCLUSIONS:
Overall, several flavonoids from P. corylifolia might be useful remedies for treating inflammatory diseases by inhibiting IL-6-induced STAT3 activation and phosphorylation. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)