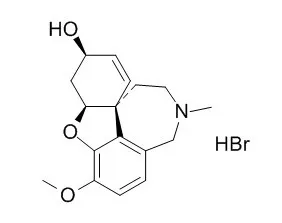
| Structure Identification: |
| J Pharm Biomed Anal. 2011 Apr 28;55(1):85-92. | | Stability-indicating study of the anti-Alzheimer's drug galantamine hydrobromide.[Pubmed: 21300511] | Galantamine hydrobromide was subjected to different stress conditions (acidic, alkaline, thermal, photolytic and oxidative). Degradation was found to occur under acidic, photolytic and oxidative conditions, while the drug was stable under alkaline and elevated temperature conditions.
METHODS AND RESULTS:
A stability-indicating reversed-phase liquid chromatographic method was developed for the determination of the drug in the presence of its degradation products. The method was validated for linearity, precision, accuracy, specificity, selectivity and intermediate precision. Additionally, the degradation kinetics of the drug was assessed in relevant cases. The kinetics followed a first order behavior in the case of acidic and photolytic degradation, while a two-phase kinetics behavior was found for the oxidative degradation.
CONCLUSIONS:
The degradation products were characterized by mass spectrometry and nuclear magnetic resonance spectroscopy.
Dehydration, epimerization and N-oxidation were the main processes observed during the degradation of galantamine. Moreover, if sufficient material could be isolated the inhibitory activity against the target enzyme acetylcholinesterase was also assessed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)