| In vitro: |
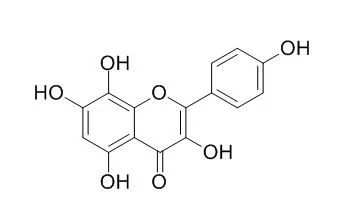
| Eur J Pharmacol. 2015 Oct 15;765:115-23. | | Herbacetin inhibits inducible nitric oxide synthase via JNK and nuclear factor-κB in LPS-stimulated RAW264.7 cells.[Pubmed: 26297979 ] | Herbacetin (3,4',5,7,8-pentahydroxyflavone), an active flavonol compound within flavonoid, has been shown to induce apoptosis in HepG2 cells and suppress hepatocyte growth factor-induced motility of human breast cancer MDA-MB-231 cells. However, the anti-inflammatory mechanisms of Herbacetin have not been researched.
METHODS AND RESULTS:
In this study, we examined the inflammatory responses stimulated by lipopolysaccharide (LPS) in RAW264.7 macrophage cells after pretreatment with different concentrations of Herbacetin. We found that Herbacetin decreased nitric oxide (NO) production in LPS-induced RAW264.7 and mouse bone marrow-derived macrophages. In addition, Herbacetin inhibited the LPS-induced expression of inducible nitric oxide synthase mRNA and protein in RAW264.7 cells. Treatment with Herbacetin decreased the release of proinflammatory cytokines, including TNF-α and IL-1β. Moreover, Herbacetin inhibited the activity of JNK kinase and nuclear factor-κB, signaling molecules involved in NO production. Cell signaling analysis using Bay 11-7082 (an inhibitory κB kinase 2 inhibitor) and mitogen-activated protein kinase (MAPK) inhibitors (SB203580 for p38, SP600125 for JNK, and PD 98059 for ERK) suggested that LPS induced iNOS expression via activation of the JNK and NF-κB pathway, but not the p38 and ERK pathway.
CONCLUSIONS:
These findings suggest that Herbacetin exerts an anti-inflammatory effect through suppression of LPS-induced JNK and NF-κB signaling pathways and diminished production of proinflammatory cytokines and mediators. | | Planta Med. 2013 Nov;79(16):1525-30. | | Herbacetin, a constituent of ephedrae herba, suppresses the HGF-induced motility of human breast cancer MDA-MB-231 cells by inhibiting c-Met and Akt phosphorylation.[Pubmed: 24081687] | Ephedrae herba suppresses hepatocyte growth factor-induced cancer cell motility by inhibiting tyrosine phosphorylation of the hepatocyte growth factor receptor, c-Met, and the PI3K/Akt pathway. Moreover, Ephedrae herba directly inhibits the tyrosine-kinase activity of c-Met. Ephedrine-type alkaloids, which are the active component of Ephedrae herba, do not affect hepatocyte growth factor-c-Met-Akt signalling, prompting us to study other active molecules in the herb.
METHODS AND RESULTS:
We recently discovered Herbacetin glycosides and found that their aglycon, Herbacetin, inhibits hepatocyte growth factor-c-Met-Akt signalling. This study revealed a novel biological activity of Herbacetin. Herbacetin suppressed hepatocyte growth factor-induced motility in human breast cancer MDA-MB-231 cells by inhibiting c-Met and Akt phosphorylation and directly inhibiting c-Met tyrosine kinase activity. The effects of Herbacetin were compared to those of kaempferol, apigenin, and isoscutellarein, all of which have similar structures. Herbacetin inhibition of hepatocyte growth factor-induced motility was the strongest of those for the tested flavonols, and only Herbacetin inhibited the hepatocyte growth factor-induced phosphorylation of c-Met.
CONCLUSIONS:
These data suggest that Herbacetin is a novel Met inhibitor with a potential utility in cancer therapeutics. | | Food Science, 2013, 34(17):106-10. | | In vitro Free Radical Scavenging and Protein Oxidation Inhibitory Effects of Herbacetin[Reference: WebLink] |
METHODS AND RESULTS:
The in vitro antioxidant activity of Herbacetin was investigated in terms of scavenging activities against DPPH and hydroxyl free radicals and inhibitory effects against oxidative protein damage and carbonylation induced by Cu 2+-H 2 O 2 or AAPH. At a concentration between 10 μ mol/L and 100 μ mol/L, Herbacetin exerted a marked scavenging effect on DPPH and hydroxyl free radicals, with respective IC 50 values of 49.28 μ mol/L and 219.2 μ mol/L. Meanwhile, Herbacetin could significantly inhibit oxidative protein damage and carbonylation induced by Cu 2+-H 2 O 2 or AAPH in a dose-dependent manner.
CONCLUSIONS:
This study concludes that Herbacetin has a strong ability to scavenge free radical and inhibit oxidative protein damage. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)