| Structure Identification: |
| Chem Pharm Bull (Tokyo). 1995 Dec;43(12):2187-94. | | Studies on the constituents of Clematis species. VI. The constituents of Clematis stans Sieb. et Zucc.[Pubmed: 8582022] |
METHODS AND RESULTS:
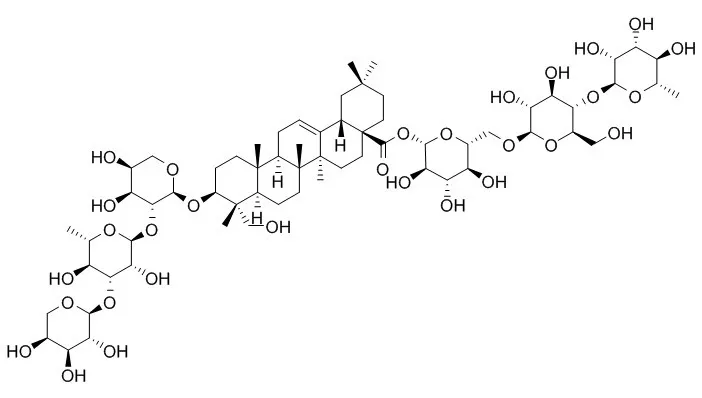
From the roots of Clematis stans three new oleanane-type triterpenoid saponins named clemastanoside A, B and C, and two new lignan glycosides named clemastanin A and B, have been isolated together with three known triterpenoid saponins, huzhangoside B, C and D, and three known lignan glycosides, (+)-lariciresinol 4-O-beta-D-glucopyranoside, (+)-lariciresinol 4'-O-beta-D-glucopyranoside and (+)-pinoresinol 4,4'-O-bis-beta-D-glucopyranoside. In addition, from the leaves, four new oleanane-type triterpenoid saponins, named clemastanoside D, E, F and G, have been isolated together with five known triterpenoid saponins, hederasaponin B, kizutasaponin K12, huzhangoside B, sieboldianoside B and Huzhangoside D, and three known flavonoids, isoquercitrin, rutin and quercetin 3-O-beta-D-glucuronopyranoside. | | Pak J Biol Sci. 2007 Jun 15;10(12):2066-72. | | Chemodiversity of saponins and their taxonomic importance in Clematis genus (Ranunculaceae).[Pubmed: 19093448 ] | Distribution patterns of chemical compounds in plants have been used for biosystematic and phylogenetic studies.
METHODS AND RESULTS:
Saponin profile of twelve major taxa of Clematis genus, belonging to sections, Rectae, Clematis, Meclatis, Tubulosae and Viorna were analyzed by HPLC coupled with diode array detector and ESI-MS. The chemodiversity profile of saponins has unambiguously delimited the taxa of Clematis at subgenus, section and subsection level. The distribution of saponins in Clematis genus provides useful taxonomic markers and results are presented in phenograms.
CONCLUSIONS:
The compound Huzhangoside D was common and the most abundant in analyzed species of the genus. The morphological analysis was also conducted of the same taxa and presented as cluster tree. The distribution and chemotaxonomic importance of saponins profile within the genus is discussed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)