| Kinase Assay: |
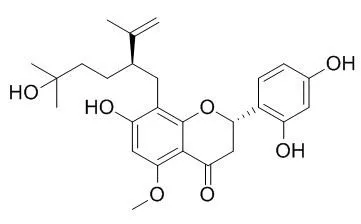
| Toxicol Appl Pharmacol. 2014 Dec 1;281(2):157-65. | | Kurarinol induces hepatocellular carcinoma cell apoptosis through suppressing cellular signal transducer and activator of transcription 3 signaling.[Pubmed: 24997323] | Kurarinol is a flavonoid isolated from roots of the medical plant Sophora flavescens. However, its cytotoxic activity against hepatocellular carcinoma (HCC) cells and toxic effects on mammalians remain largely unexplored.
METHODS AND RESULTS:
Here, the pro-apoptotic activities of Kurarinol on HCC cells and its toxic impacts on tumor-bearing mice were evaluated. The molecular mechanisms underlying Kurarinol-induced HCC cell apoptosis were also investigated. We found that Kurarinol dose-dependently provoked HepG2, Huh-7 and H22 HCC cell apoptosis. In addition, Kurarinol gave rise to a considerable decrease in the transcriptional activity of signal transducer and activator of transcription 3 (STAT3) in HCC cells. Suppression of STAT3 signaling is involved in Kurarinol-induced HCC cell apoptosis. In vivo studies showed that Kurarinol injection substantially induced transplanted H22 cell apoptosis with low toxic impacts on tumor-bearing mice. Similarly, the transcriptional activity of STAT3 in transplanted tumor tissues was significantly suppressed after Kurarinol treatment.
CONCLUSIONS:
Collectively, our current research demonstrated that Kurarinol has the capacity of inducing HCC cell apoptosis both in vitro and in vivo with undetectable toxic impacts on the host. Suppressing STAT3 signaling is implicated in Kurarinol-mediated HCC cell apoptosis. | | Biol Pharm Bull. 2008 Jan;31(1):154-8. | | Inhibitory effects of kurarinol, kuraridinol, and trifolirhizin from Sophora flavescens on tyrosinase and melanin synthesis.[Pubmed: 18175961 ] | Previously, it was reported that some prenylated flavonoids contained in the dichloromethane fraction of the ethanolic extract of Sophora flavescens, such as kuraridin, sophoraflavanone G, kurarinone, and kushenol F, are tyrosinase inhibitors; however, based on the level of these inhibitors in the extract, its inhibitory effect on tyrosinase activity was higher than expected. This has led us to further investigate other possible constituents that may contribute to the extract's strong inhibitory activity.
METHODS AND RESULTS:
The results of this study indicate that Kurarinol (1), kuraridinol (2), and trifolirhizin (3), from the ethyl acetate fraction of Sophora extract, can inhibit tyrosinase activity. Compared with kojic acid (16.22+/-1.71 microM), compounds 1-3 possessed potent tyrosinase inhibitory activity with IC(50) values of 8.60+/-0.51, 0.88+/-0.06, and 506.77+/-4.94 microM, respectively. These three compounds were further tested for their inhibitory effects on melanogenesis. In cultured B16 melanoma cells, 1-3 markedly inhibited (>50%) melanin synthesis at 50 microM.
CONCLUSIONS:
This is the first study indicating that 1-3 exert varying degrees of inhibition on tyrosinase-dependent melanin biosynthesis, and therefore, are candidates as skin-whitening agents. | | Phytomedicine. 2008 Aug;15(8):612-8. Epub 2007 Oct 24. | | Kurarinol, tyrosinase inhibitor isolated from the root of Sophora flavescens.[Pubmed: 17951038] | It is well known that flavanones, sophoraflavanone G 1, kurarinone 2, and Kurarinol 3, from the root of Sophora flavescens, have extremely strong tyrosinase inhibitory activity.
METHODS AND RESULTS:
This study delineates the principal pharmacological features of Kurarinol 3 that lead to inhibition of the oxidation of l-tyrosine to melanin by mushroom tyrosinase (IC(50) of 100 nM). The inhibition kinetics analyses unveil that compounds 1 and 2 are noncompetitive inhibitors. However similar analysis shows Kurarinol 3 to be a competitive inhibitor. Compounds 1 and 2 exhibited potent antibacterial activity with 10 microg/disk against Gram-positive bacteria, whereas Kurarinol 3 did not ostend any antibacterial activity. Interestingly, Kurarinol 3 inhibits production of melanin in S. bikiniensis without affecting the growth of microorganism. It is thus distinctly different from the other tyrosinase inhibitors 1 and 2. In addition, Kurarinol 3 manifests relatively low cytotoxic activity (EC(50)>30 microM) compared to 1 and 2. To account for these observations, we conducted molecular modeling studies.
CONCLUSIONS:
These suggested that the lavandulyl group within Kurarinol is instrumental in the interaction with the enzyme. More specifically, the terminal hydroxy function within the lavandulyl group is most important for optimal binding. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)