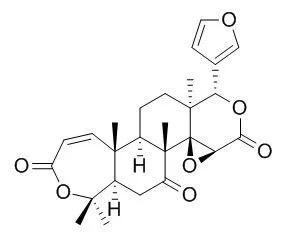
| Description: |
Obacunone is a novel activator of Nrf2, which exhibits anti-cancer, anti-inflammatory, antivirulence, insecticidal, anti-proliferative and anti-aromatase activities. Obacunone stimulates muscle hypertrophy and prevents obesity and hyperglycemia, and that these beneficial effects are likely to be mediated through the activation of TGR5 and inhibition of PPARγ transcriptional activity. Obacunone significantly inhibits aromatase activity in an in vitro enzyme assay with an IC50 value of 28.04 μM, and it also inhibits the p38 MAPK signaling pathway. |
| Targets: |
Caspase | Akt | p38MAPK | PPAR | Nrf2 | P450 (e.g. CYP17) | Bcl-2/Bax | NF-kB | COX | Antifection | TGR5 |
| In vitro: |
| Toxicology. 2015 Mar 2;329:88-97. | | Cytotoxicity of obacunone and obacunone glucoside in human prostate cancer cells involves Akt-mediated programmed cell death.[Pubmed: 25592883] | Obacunone and Obacunone glucoside (OG) are naturally occurring triterpenoids commonly found in citrus and other plants of the Rutaceae family.
The current study reports the mechanism of cytotoxicity of citrus-derived Obacunone and OG on human androgen-dependent prostate cancer LNCaP cells.
METHODS AND RESULTS:
Both limonoids exhibited time- and dose-dependent inhibition of cell proliferation, with more than 60% inhibition of cell viability at 100 μM, after 24 and 48 h. Analysis of fragmentation of DNA, activity of caspase-3, and cytosolic cytochrome-c in the cells treated with limonoids provided evidence for activation of programmed cell death by limonoids. Treatment of LNCaP cells with Obacunone and OG resulted in dose-dependent changes in expression of proteins responsible for the induction of programmed cell death through the intrinsic pathway and down-regulation of Akt, a key molecule in cell signaling pathways. In addition, Obacunone and OG also negatively regulated an inflammation-associated transcription factor, androgen receptor, and prostate-specific antigen, and activated proteins related to the cell cycle, confirming the ability of limonoids to induce cytotoxicity through multiple pathways.
CONCLUSIONS:
The results of this study provided, for the first time, an evidence of the cytotoxicity of Obacunone and OG in androgen-dependent human prostate cancer cells. |
|
| In vivo: |
| Appl Environ Microbiol. 2012 Oct;78(19):7012-22. | | Obacunone represses Salmonella pathogenicity islands 1 and 2 in an envZ-dependent fashion.[Pubmed: 22843534] | Obacunone belongs to a class of unique triterpenoids called limonoids, present in Citrus species. Previous studies from our laboratory suggested that Obacunone possesses antivirulence activity and demonstrates inhibition of cell-cell signaling in Vibrio harveyi and Escherichia coli O157:H7.
METHODS AND RESULTS:
The present work sought to determine the effect of Obacunone on the food-borne pathogen Salmonella enterica serovar Typhimurium LT2 by using a cDNA microarray.
Transcriptomic studies indicated that Obacunone represses Salmonella pathogenicity island 1 (SPI1), the maltose transporter, and the hydrogenase operon. Furthermore, phenotypic data for the Caco-2 infection assay and maltose utilization were in agreement with microarray data suggesting repression of SPI1 and maltose transport. Further studies demonstrated that repression of SPI1 was plausibly mediated through hilA. Additionally, Obacunone seems to repress SPI2 under SPI2-inducing conditions as well as in Caco-2 infection models. Furthermore, Obacunone seems to repress hilA in an EnvZ-dependent fashion.
CONCLUSIONS:
Altogether, the results of the study seems to suggest that Obacunone exerts an antivirulence effect on S. Typhimurium and may serve as a lead compound for development of antivirulence strategies for S. Typhimurium. | | Biochem Biophys Res Commun. 2015 Aug 7;463(4):846-52. | | Dietary obacunone supplementation stimulates muscle hypertrophy, and suppresses hyperglycemia and obesity through the TGR5 and PPARγ pathway.[Pubmed: 26051277 ] | Obacunone is a limonoid that is predominantly found in Citrus. Although various biological activities of limonoids have been reported, little is known about the beneficial effects of Obacunone on metabolic disorders. In the present study, we examined the effects of dietary Obacunone supplementation on obese KKAy mice, to clarify the function of Obacunone in metabolic regulation.
METHODS AND RESULTS:
Mice were pair-fed a normal diet either alone or supplemented with 0.1% w/w Obacunone for 28 days. Compared with the control, Obacunone-fed mice had lower glycosylated hemoglobin, blood glucose, and white adipose tissue weight, although there was no significant difference in body weight. Obacunone treatment also significantly increased the weight of the gastrocnemius and quadriceps muscles. Reporter gene assays revealed that Obacunone stimulated the transcriptional activity of the bile acids-specific G protein-coupled receptor, TGR5, in a dose-dependent manner. In addition, Obacunone inhibited adipocyte differentiation in 3T3-L1 cells and antagonized ligand-stimulated peroxisome proliferator-activated receptor γ (PPARγ) transcriptional activity.
CONCLUSIONS:
These results suggest that Obacunone stimulates muscle hypertrophy and prevents obesity and hyperglycemia, and that these beneficial effects are likely to be mediated through the activation of TGR5 and inhibition of PPARγ transcriptional activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)