| In vivo: |
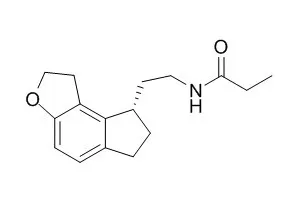
| Arch Gen Psychiatry. 2006 Oct;63(10):1149-57. | | Ramelteon: a novel hypnotic lacking abuse liability and sedative adverse effects.[Pubmed: 17015817 ] | Ramelteon is a novel MT1 and MT2 melatonin receptor selective agonist recently approved for insomnia treatment. Most approved insomnia medications have potential for abuse and cause motor and cognitive impairment.
To evaluate the potential for abuse, subjective effects, and motor and cognitive-impairing effects of Ramelteon compared with triazolam, a classic benzodiazepine sedative-hypnotic drug.
METHODS AND RESULTS:
In this double-blind crossover study, each participant received oral doses of Ramelteon (16, 80, or 160 mg), triazolam (0.25, 0.5, or 0.75 mg), and placebo during approximately 18 days. All participants received each treatment on different days. Most outcome measures were assessed at 0.5 hours before drug administration and repeatedly up to 24 hours after drug administration.SETTING:
Residential research facility.PARTICIPANTS:
Fourteen adults with histories of sedative abuse.
Subject-rated measures included items relevant to potential for abuse (eg, drug liking, street value, and pharmacological classification), as well as assessments of a broad range of stimulant and sedative subjective effects. Observer-rated measures included assessments of sedation and impairment. Motor and cognitive performance measures included psychomotor and memory tasks and a standing balance task.
Compared with placebo, Ramelteon (16, 80, and 160 mg) showed no significant effect on any of the subjective effect measures, including those related to potential for abuse. In the pharmacological classification, 79% (11/14) of subjects identified the highest dose of Ramelteon as placebo. Similarly, compared with placebo, Ramelteon had no effect at any dose on any observer-rated or motor and cognitive performance measure. In contrast, triazolam showed dose-related effects on a wide range of subject-rated, observer-rated, and motor and cognitive performance measures, consistent with its profile as a sedative drug with abuse liability.
CONCLUSIONS:
Ramelteon demonstrated no significant effects indicative of potential for abuse or motor and cognitive impairment at up to 20 times the recommended therapeutic dose and may represent a useful alternative to existing insomnia medications. | | J Clin Gastroenterol. 2015 Apr 9. | | The Effect of Ramelteon on Heartburn Symptoms of Patients With Gastroesophageal Reflux Disease and Chronic Insomnia: A Pilot Study.[Pubmed: 25887111] | There is a bidirectional relationship between gastroesophageal reflux disease (GERD) and sleep. It has been demonstrated that antireflux treatment can improve sleep quality in GERD patients with nighttime reflux.
METHODS AND RESULTS:
Patients with heartburn and/or regurgitation ≥3 times/week and insomnia for ≥3 months were included. Patients were assessed at baseline with the demographic, GERD symptom assessment scale, Epworth sleepiness scale, Berlin sleep apnea, Pittsburgh sleep quality index, and the Insomnia severity index questionnaires. Subjects underwent an upper endoscopy followed by pH testing. Subsequently, subjects were randomized, in a double-blind, placebo-controlled trial, to receive either Ramelteon 8 mg or placebo before bedtime for 4 weeks. During the last week of treatment, subjects completed a daily GERD symptom and sleep diary and underwent actigraphy. Sixteen patients completed the study, 8 in each arm (mean age and M/F were 48.5 vs. 57.8 y, and 8/0 vs. 6/2, respectively). Patients who received Ramelteon demonstrated a statistically significant decrease in symptom score as compared with those who received placebo for daytime heartburn (-42% vs. -29%), nighttime heartburn (-42% vs. 78%), 24-hour heartburn (-42% vs. -3%), and 24-hour acid regurgitation (-26% vs. 19%) (all P<0.05). Insomnia severity index score was significantly reduced in patients receiving Ramelteon as compared with placebo (-46% vs. -5%, P<0.05). Ramelteon group also demonstrated a significant improvement in sleep efficiency and sleep latency, as compared with placebo, P<0.05). No significant adverse events were observed with Ramelteon.
CONCLUSIONS:
Ramelteon significantly improved symptoms in patients with GERD. In addition, Ramelteon significantly improved patients' sleep experience. Further studies are needed in the future (NCT01128582). | | Clin Psychopharmacol Neurosci. 2014 Dec;12(3):215-7. | | Effects of Add-on Ramelteon on Cognitive Impairment in Patients with Schizophrenia: An Open-label Pilot Trial.[Pubmed: 25598825] | This open-label study examined the effects of Ramelteon on cognitive functions in 10 outpatients with schizophrenia.
METHODS AND RESULTS:
Ramelteon (8 mg/day) was administered to 10 patients with schizophrenia for six months. The verbal fluency test, Trail-Making Test, the Wisconsin Card Sorting Test, the Stroop Test, the Digit Span Distraction Test, Iowa Gambling Task, the Rey Auditory Verbal Learning Test were evaluated at baseline and 6 months after treatment with Ramelteon.
Ramelteon improved significantly the scores of Rey Auditory Verbal Learning Test. Additionally, Ramelteon exerted improvements in the verbal fluency and Iowa Gambling Task in 4 patients.
CONCLUSIONS:
Ramelteon could be a potential therapeutic drug, in adjunctive treatment of learning and memory deficits seen in patients with schizophrenia. | | JAMA Psychiatry. 2014 Apr;71(4):397-403. | | Preventive effects of ramelteon on delirium: a randomized placebo-controlled trial.[Pubmed: 24554232] | No highly effective interventions to prevent delirium have been identified. To examine whether Ramelteon, a melatonin agonist, is effective for the prevention of delirium. DESIGN, SETTING, AND PARTICIPANTS: A multicenter, rater-blinded, randomized placebo-controlled trial was performed in intensive care units and regular acute wards of 4 university hospitals and 1 general hospital. Eligible patients were 65 to 89 years old, newly admitted due to serious medical problems, and able to take medicine orally. Patients were excluded from the study if they had an expected stay or life expectancy of less than 48 hours.
METHODS AND RESULTS:
Sixty-seven patients were randomly assigned using the sealed envelope method to receive Ramelteon (8 mg/d; 33 patients) or placebo (34 patients) every night for 7 days. Incidence of delirium, as defined by the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition). Ramelteon was associated with a lower risk of delirium (3% vs 32%; P = .003), with a relative risk of 0.09 (95% CI, 0.01-0.69). Even after risk factors were controlled for, Ramelteon was still associated with a lower incidence of delirium (P = .01; odds ratio, 0.07 [95% CI, 0.008-0.54]). The Kaplan-Meier estimates of time to development of delirium were 6.94 (95% CI, 6.82-7.06) days for Ramelteon and 5.74 (5.05-6.42) days for placebo. Comparison by log-rank test showed that the frequency of delirium was significantly lower in patients taking Ramelteon than in those taking placebo (χ(2) = 9.83; P = .002).
CONCLUSIONS:
Ramelteon administered nightly to elderly patients admitted for acute care may provide protection against delirium. This finding supports a possible pathogenic role of melatonin neurotransmission in delirium. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)