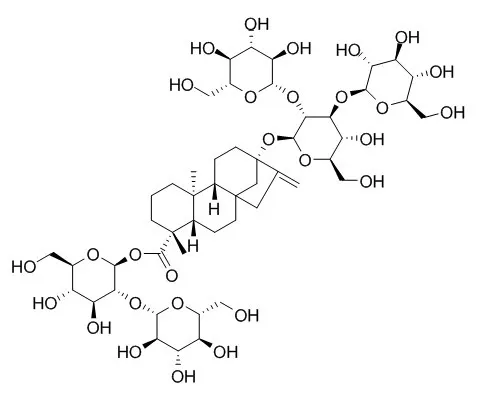
| Structure Identification: |
| Food Chem. 2015 May 1;174:564-70. | | Investigation of the solubility enhancement mechanism of rebaudioside D using a solid dispersion technique with potassium sorbate as a carrier.[Pubmed: 25529720] | Rebaudioside (Reb) D is a high intensity, natural sweetener that shows great potential for substituting sugar in sweetened beverages. However, Rebaudioside D is poorly water soluble, and thus, a solid dispersion technique was recently established to enhance its solubility.
METHODS AND RESULTS:
The purpose of this study was to elucidate the solubility enhancement mechanism of this solid dispersion material by employing Scanning Electron Microscopy (SEM), Raman spectroscopy, Fourier Transform Infrared spectroscopy (FT-IR) and X-ray Diffraction (XRD). Potassium sorbate (KS) was chosen as the carrier and two different concentration ratios were investigated as solid dispersions (SD) and as physical mixtures (PM).
CONCLUSIONS:
Our data demonstrated the possible mechanism for enhancing solubility through solid dispersion through increased surface area/volume ratio and hydrogen bonding between Rebaudioside D and KS. The interaction between the two components were also related to the different concentration ratios, therefore an optimisation of the ratio is important to produce a soluble and stable complex. | | Int J Mol Sci. 2012 Nov 16;13(11):15126-36. | | Catalytic hydrogenation of the sweet principles of Stevia rebaudiana, Rebaudioside B, Rebaudioside C, and Rebaudioside D and sensory evaluation of their reduced derivatives.[Pubmed: 23203115] | Catalytic hydrogenation of rebaudioside B, rebaudioside C, and Rebaudioside D; the three ent-kaurane diterpene glycosides isolated from Stevia rebaudiana was carried out using Pd(OH)(2).
METHODS AND RESULTS:
Reduction of steviol glycosides was performed using straightforward synthetic chemistry with the catalyst Pd(OH)(2) and structures of the corresponding dihydro derivatives were characterized on the basis of 1D and 2D nuclear magnetic resonance (NMR) spectral data indicating that all are novel compounds being reported for the first time. Also, the taste properties of all reduced compounds were evaluated against their corresponding original steviol glycosides and sucrose. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)