| Description: |
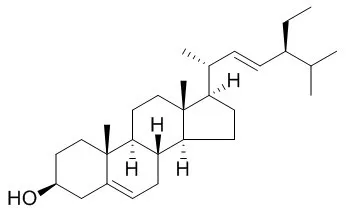
Stigmasterol is used as a precursor in the manufacture of synthetic progesterone, it is an antagonist of the bile acid nuclear receptor FXR, which has anti-inflammatory, thyroid inhibitory, cholesterol-lowering, antiperoxidative and hypoglycemic effects; it has indicated that stigmasterol may be useful in prevention of certain cancers, including ovarian, prostate, breast, and colon cancers. Stigmasterol inhibits the NF-kappaB pathway. |
| Targets: |
IL Receptor | MMP(e.g.TIMP) | PGE | NF-kB | Bcl-2/Bax | Caspase | p53 | ERK | cAMP | NMDAR | SOD |
| In vitro: |
| Osteoarthritis Cartilage. 2010 Jan;18(1):106-16. | | Stigmasterol: a phytosterol with potential anti-osteoarthritic properties.[Pubmed: 19786147] | Although most studies have focused on the cholesterol-lowering activity of Stigmasterol, other bioactivities have been ascribed to this plant sterol compound, one of which is a potential anti-inflammatory effect. To investigate the effects of Stigmasterol, a plant sterol, on the inflammatory mediators and metalloproteinases produced by chondrocytes.
METHODS AND RESULTS:
We used a model of newborn mouse chondrocytes and human osteoarthritis (OA) chondrocytes in primary culture stimulated with or without IL-1beta (10 ng/ml), for 18 h. Cells were pre-incubated for 48 h with Stigmasterol (20 microg/ml) compared to untreated cells. We initially investigated the presence of Stigmasterol in chondrocyte, compared to other phytosterols. We then assessed the role of Stigmasterol on the expression of various genes involved in inflammation (IL-6) and cartilage turn-over (MMP-3, -13, ADAMTS-4, -5, type II collagen, aggrecan) by quantitative Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR). Additional experiments were carried out to monitor the production of MMP-3 and prostaglandin E2 (PGE(2)) by specific immuno-enzymatic assays. We eventually looked at the role of Stigmasterol on NF-kappaB activation by western blot, using an anti-IkappaBalpha antibody.
After 18 h of IL-1beta treatment, MMP-3, MMP-13, ADAMTS-4, but not ADAMTS-5 RNA expression were elevated, as well as MMP-3 and PGE(2) protein levels in mouse and human chondrocytes. Type II collagen and aggrecan mRNA levels were significatively reduced. Pre-incubation of Stigmasterol to IL-1beta-treated cells significantly decreased these effects described above (significant reduction of MMP-3 mRNA in human and mouse, MMP-3 protein in mouse, MMP-13 mRNA in mouse and human, ADAMTS-4 mRNA in human, PGE(2) protein in human and mouse) Finally, Stigmasterol was capable of counteracting the IL-1beta-induced NF-kappaB pathway.
CONCLUSIONS:
This study shows that Stigmasterol inhibits several pro-inflammatory and matrix degradation mediators typically involved in OA-induced cartilage degradation, at least in part through the inhibition of the NF-kappaB pathway. These promising results justify further ex vivo and in vivo investigations with Stigmasterol. |
|
| In vivo: |
| Eur J Pharmacol. 2012 Feb 15;676(1-3):64-70. | | The ameliorating effects of stigmasterol on scopolamine-induced memory impairments in mice.[Pubmed: 22173129] | Stigmasterol, a kind of phytosterol, is present in small amounts in various foods.
METHODS AND RESULTS:
In the present study, we investigated the effects of Stigmasterol on scopolamine-induced memory impairments using the passive avoidance and the Morris water maze tasks in mice. In addition, changes in memory-related molecules, including extracellular signal-regulated kinase (ERK) and cAMP response element-binding protein (CREB), were examined following the administration of Stigmasterol. Scopolamine-induced memory impairments were significantly attenuated by the administration of Stigmasterol (10mg/kg) in the passive avoidance task. In the Morris water maze task, the escape latencies were significantly decreased in the Stigmasterol-treated group compared to the scopolamine-treated group during the training phase. The swimming times within the target zone during the probe trial were significantly increased as compared to scopolamine-treated mice. Furthermore, the ameliorating effect of Stigmasterol on scopolamine-induced memory dysfunction was blocked by a sub-effective dose of dizocilpine (MK-801), an NMDA receptor antagonist, and tamoxifen, an estrogen receptor antagonist, in the passive avoidance task. In addition, the expression levels of phosphorylated ERK and CREB in the hippocampus were significantly increased by Stigmasterol, which was blocked by tamoxifen or MK-801 with scopolamine.
CONCLUSIONS:
These results suggest that Stigmasterol-induced cognitive ameliorative effects are mediated by the enhancement of cholinergic neurotransmission system via the activation of estrogen or NMDA receptors. | | Metabolism. 2006 Mar;55(3):292-9. | | Stigmasterol reduces plasma cholesterol levels and inhibits hepatic synthesis and intestinal absorption in the rat.[Pubmed: 16483871] | Plant sterols compete with cholesterol (cholest-5-en-3beta-ol) for intestinal absorption to limit absorption and lower plasma concentrations of cholesterol. Stigmasterol (24-ethyl-cholesta-5,22-dien-3beta-ol; Delta(22) derivative of sitosterol [24-ethyl-cholest-5-en-3beta-ol]), but not campesterol (24-methyl-cholest-5-en-3beta-ol) and sitosterol, is reported to inhibit cholesterol biosynthesis via inhibition of sterol Delta(24)-reductase in human Caco-2 and HL-60 cell lines.
METHODS AND RESULTS:
We studied the effect of feeding 0.5% Stigmasterol on plasma and liver sterols and intestinal cholesterol and sitosterol absorption in 12 wild-type Kyoto (WKY) and 12 Wistar rats. After 3 weeks of feeding, cholesterol and sitosterol absorption was determined in 6 rats from each group by plasma dual-isotope ratio method. After 3 more weeks, plasma and hepatic sterols and hepatic enzyme activities were determined in all rats. After feeding Stigmasterol, baseline plasma cholesterol was 1.3 times and plant sterols 3 times greater in WKY compared with Wistar rats. Stigmasterol feeding lowered plasma cholesterol by approximately 11%, whereas plasma campesterol and sitosterol levels were virtually unchanged in both rat strains, and Stigmasterol constituted 3.2% of plasma sterols in WKY rats and 1% in Wistar rats. After 6 weeks of feeding, cholesterol and sitosterol absorption decreased 23% and 30%, respectively, in WKY, and 22% and 16%, respectively, in the Wistar rats as compared with untreated rats. The intestinal bacteria in both rat strains metabolized Stigmasterol to mainly the 5beta-H stanol (>40%), with only small amounts of 5alpha-H derivative (approximately 1.5%), whereas the C-22 double bond was resistant to bacterial metabolism. Hepatic Stigmasterol levels increased from 11 microg/g liver tissue to 104 mug/g in WKY rats and from 5 microg/g liver tissue to 21 microg/g in Wistar rats. 3-Hydroxy-3-methylglutaryl coenzyme A reductase activity was suppressed 4-fold in the WKY and almost 1.8-fold in Wistar rats, cholesterol 7alpha-hydroxylase activity was suppressed 1.6-fold in the WKY and 3.5-fold in Wistar rats, whereas cholesterol 27-hydroxylase activity was unchanged after feeding.
CONCLUSIONS:
In conclusion, Stigmasterol, when fed, lowers plasma cholesterol levels, inhibits intestinal cholesterol and plant sterol absorption, and suppresses hepatic cholesterol and classic bile acid synthesis in Wistar as well as WKY rats. However, plasma and hepatic incorporation of Stigmasterol is low. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)