| Description: |
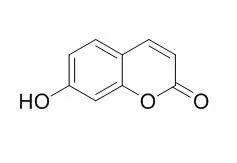
Umbelliferone is a competitive inhibitor of alkaline phosphatase. It is a fluorescing compound used as a sunscreen agent and shows good inhibitions of DPPH, hydroxyl, superoxide anion and ABTS radicals with antinociceptive, anti-inflammatory, anti-hyperglycaemic, anti-allergic, molluscicidal and anti-tumor activities. Umbelliferone has stimulatory effect on adipocyte differentiation likely occurs through up-regulation of adipogenic transcription factors and downstream adipocyte-specific gene expression.
|
| Targets: |
P450 (e.g. CYP17) | 5-alpha Reductase | IFN-γ | IL Receptor |
| In vitro: |
| Mol Med Rep. 2015 May 18. | | Umbelliferone exhibits anticancer activity via the induction of apoptosis and cell cycle arrest in HepG2 hepatocellular carcinoma cells.[Pubmed: 25997538] | Hepatocellular carcinoma (HCC) is a highly malignant tumor, associated with poor patient prognoses, and high rates of morbidity and mortality. To date, the therapeutic strategies available for the treatment of HCC remain limited.
The present study aimed to elucidate the anticancer activity of Umbelliferone, a naturally occurring coumarin derivative isolated from Ferula communis, against the HepG2 HCC cell line.
METHODS AND RESULTS:
A 3‑(4,5‑dimthylthaizol‑2‑yl)‑2,5, diphenyltetrazolium bromide assay was used to evaluate cell viability following Umbelliferone treatment, and the effects of Umbelliferone on cell cycle progression and apoptosis were evaluated using flow cytometry. The presence of morphological features characteristic of apoptosis, including cell shrinkage, membrane blebbing, nuclear condensation and apoptotic body formation, were evaluated in HepG2 cells following Umbelliferone treatment. Cell cycle analysis conducted via propidium iodide (PI) staining indicated that Umbelliferone treatment induced cell cycle arrest at S phase in HepG2 cells. Analysis with Annexin V and PI staining revealed that Umbelliferone induced apoptotic events in HepG2 cells in a concentration‑dependant manner (0‑50 μM). Umbelliferone also induced dose‑dependant DNA fragmentation.
CONCLUSIONS:
In conclusion, Umbelliferone was found to exhibit significant anticancer effects via the induction of apoptosis, cell cycle arrest and DNA fragmentation in HepG2 cancer cells. | | Food Chem., 2010, 120(3):825-30. | | Umbelliferone - An antioxidant isolated from Acacia nilotica (L.) Willd. Ex. Del.[Reference: WebLink] | The bark and leaves of Acacia nilotica are consumed for their promising medicinal properties in several parts of the world. The aerial portion, including flowers, is used as fodder for animals. This study aimed to isolate the functional components of A. nilotica and to check their antioxidant activities in vitro.
METHODS AND RESULTS:
In the fractionation of methanol extract, a fraction, AN-2, was isolated, which was identified by spectroscopic techniques, namely NMR and mass spectroscopy to be a coumarin derivative, i.e. Umbelliferone. The antioxidative activities, including the DPPH, deoxyribose (site and non-site specific), chelating power, reducing power and lipid peroxidation assays, were studied in vitro and performed in the Dept. of Botanical and Env. Sciences, GNDU, Amritsar. It was found that the antioxidative effect of Umbelliferone was dose-dependent up to 100 μg/ml and then levelled off with no further increase in activity.
CONCLUSIONS:
This is the first report of the isolation and antioxidant potential of Umbelliferone from A. nilotica. |
|
| In vivo: |
| Nat Prod Res. 2014;28(17):1371-4. | | In vivo antinociceptive and anti-inflammatory activities of umbelliferone isolated from Potentilla evestita.[Pubmed: 24673335] | This study was designed to evaluate the antinociceptive and anti-inflammatory activities of a compound, Umbelliferone, isolated from the chloroform fraction of Potentilla evestita in animal models.
METHODS AND RESULTS:
When tested against acetic acid-induced noxious stimulus, it significantly prolonged pain threshold and provided 38.38% and 60.95% reduction in abdominal constriction at 5 and 10 mg/kg i.p., respectively. Post-Umbelliferone injection evoked significant dose-dependent reduction in noxious stimulation with 33.65% and 58.89% pain attenuation at 5 and 10 mg/kg i.p., respectively, in the initial phase. In the late phase, it illustrated more dominant effect with 37.65% and 63.79% blockade of painful sensation. Similarly, it exhibited significant anti-inflammatory activity during various assessment times (1-5 h) with 46.28% and 66.13% amelioration after 4th of administration against induced oedema.
CONCLUSIONS:
In conclusion, Umbelliferone possessed strong antinociceptive and anti-inflammatory activities by inhibiting both peripheral and centrally acting pain mediators. | | Chem Biol Interact. 2014 Jun 5;216:9-16. | | Long-term supplementation of umbelliferone and 4-methylumbelliferone alleviates high-fat diet induced hypertriglyceridemia and hyperglycemia in mice.[Pubmed: 24661945] | This study was conducted to evaluate the effects of Umbelliferone (UF) and 4-methylUmbelliferone (mUF) on high-fat diet-induced hypertriglyceridemia and hyperglycemia in mice.
METHODS AND RESULTS:
The mice were assigned to normal control, high-fat control, and high-fat with UF or mUF groups. For UF or mUF groups, the high-fat diet was supplemented with UF or mUF at 0.02% (wt/wt) for 12weeks. Both UF and mUF significantly decreased plasma triglyceride, free fatty acid and glucose levels, adipocyte size, white adipose tissue weights, and hepatic phosphatidate phosphohydrolase activity and significantly increased plasma adiponectin levels and hepatic fatty acid β-oxidation activity compared with the high-fat control group. UF and mUF improved glucose intolerance and hepatic steatosis in the high-fat fed mice. Long-term high-fat diet intake induced an increase in hepatic CYP2E1 activity and lipid peroxide and cytosolic hydrogen peroxide contents and suppressed superoxide dismutase and glutathione peroxidase activities, which were reversed by UF and mUF supplementation.
CONCLUSIONS:
These results indicate that UF and mUF similarly ameliorate hypertriglyceridemia and hyperglycemia partly by modulating hepatic lipid metabolism and the antioxidant defense system along with increasing adiponectin levels. | | Mol Med Rep . 2015 Sep;12(3):3869-3873. | | Umbelliferone exhibits anticancer activity via the induction of apoptosis and cell cycle arrest in HepG2 hepatocellular carcinoma cells[Pubmed: 25997538] | | Abstract
Hepatocellular carcinoma (HCC) is a highly malignant tumor, associated with poor patient prognoses, and high rates of morbidity and mortality. To date, the therapeutic strategies available for the treatment of HCC remain limited. The present study aimed to elucidate the anticancer activity of Umbelliferone, a naturally occurring coumarin derivative isolated from Ferula communis, against the HepG2 HCC cell line. A 3‑(4,5‑dimthylthaizol‑2‑yl)‑2,5, diphenyltetrazolium bromide assay was used to evaluate cell viability following Umbelliferone treatment, and the effects of Umbelliferone on cell cycle progression and apoptosis were evaluated using flow cytometry. The presence of morphological features characteristic of apoptosis, including cell shrinkage, membrane blebbing, nuclear condensation and apoptotic body formation, were evaluated in HepG2 cells following Umbelliferone treatment. Cell cycle analysis conducted via propidium iodide (PI) staining indicated that Umbelliferone treatment induced cell cycle arrest at S phase in HepG2 cells. Analysis with Annexin V and PI staining revealed that Umbelliferone induced apoptotic events in HepG2 cells in a concentration‑dependant manner (0‑50 μM). Umbelliferone also induced dose‑dependant DNA fragmentation. In conclusion, Umbelliferone was found to exhibit significant anticancer effects via the induction of apoptosis, cell cycle arrest and DNA fragmentation in HepG2 cancer cells. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)