| Kinase Assay: |
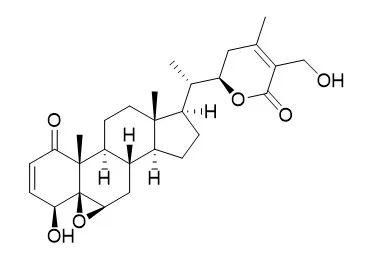
| Exp Cell Res. 2013 Nov 1;319(18):2822-34. | | Production of reactive oxygen species by withaferin A causes loss of type collagen expression and COX-2 expression through the PI3K/Akt, p38, and JNK pathways in rabbit articular chondrocytes.[Pubmed: 24016823] | Withaferin A (WFA) is a major chemical constituent of Withania somnifera, also known as Indian ginseng. Many recent reports have provided evidence of its anti-tumor, anti-inflammation, anti-oxidant, and immune modulatory activities. Although the compound appears to have a large number of effects, its defined mechanisms of action have not yet been determined.
METHODS AND RESULTS:
We investigated the effects of WFA on loss of type collagen expression and inflammation in rabbit articular chondrocytes. WFA increased the production of reactive oxygen species, suggesting the induction of oxidative stress, in a dose-dependent manner. Also, we confirmed that WFA causes loss of type collagen expression and inflammation as determined by a decrease of type II collagen expression and an increase of cyclooxygenase-2 (COX-2) expression via western blot analysis in a dose- and time- dependent manner. WFA also reduced the synthesis of sulfated proteoglycan via Alcian blue staining and caused the synthesis of prostaglandin E2 (PGE2) via assay kit in dose- and time-dependent manners. Treatment with N-acetyl-L-cysteine (NAC), an antioxidant, inhibited WFA-induced loss of type II collagen expression and increase in COX-2 expression, accompanied by inhibition of reactive oxygen species production. WFA increased phosphorylation of both Akt and p38. Inhibition of PI3K/Akt, p38, and JNK with LY294002 (LY), SB203580 (SB), or SP600125 (SP) in WFA-treated cells rescued the expression of type II collagen and suppressed the expression of COX-2.
CONCLUSIONS:
These results demonstrate that WFA induces loss of type collagen expression and inflammation via PI3K/Akt, p38, and JNK by generating reactive oxygen species in rabbit articular chondrocytes. | | Cell Death & Disease, 2013, 4(8):e778. | | Withaferin A: A proteasomal inhibitor promotes healing after injury and exerts anabolic effect on osteoporotic bone[Reference: WebLink] | Withania somnifera or Ashwagandha is a medicinal herb of Ayurveda. Though the extract and purified molecules, withanolides, from this plant have been shown to have different pharmacological activities, their effect on bone formation has not been studied.
METHODS AND RESULTS:
Here, we show that one of the withanolide, Withaferin A (WFA) acts as a proteasomal inhibitor (PI) and binds to specific catalytic β subunit of the 20S proteasome. It exerts positive effect on osteoblast by increasing osteoblast proliferation and differentiation. WFA increased expression of osteoblast-specific transcription factor and mineralizing genes, promoted osteoblast survival and suppressed inflammatory cytokines. In osteoclast, WFA treatment decreased osteoclast number directly by decreasing expression of tartarate-resistant acid phosphatase and receptor activator of nuclear factor kappa-B (RANK) and indirectly by decreasing osteoprotegrin/RANK ligand ratio. Our data show that in vitro treatment of WFA to calvarial osteoblast cells decreased expression of E3 ubiquitin ligase, Smad ubiquitin regulatory factor 2 (Smurf2), preventing degradation of Runt-related transcription factor 2 (RunX2) and relevant Smad proteins, which are phosphorylated by bone morphogenetic protein 2. Increased Smurf2 expression due to exogenous treatment of tumor necrosis factor α (TNFα) to primary osteoblast cells was decreased by WFA treatment. This was corroborated by using small interfering RNA against Smurf2. Further, WFA also blocked nuclear factor kappa-B (NF-kB) signaling as assessed by tumor necrosis factor stimulated nuclear translocation of p65-subunit of NF-kB. Overall data show that in vitro proteasome inhibition by WFA simultaneously promoted osteoblastogenesis by stabilizing RunX2 and suppressed osteoclast differentiation, by inhibiting osteoclastogenesis. Oral administration of WFA to osteopenic ovariectomized mice increased osteoprogenitor cells in the bone marrow and increased expression of osteogenic genes. WFA supplementation improved trabecular micro-architecture of the long bones, increased biomechanical strength parameters of the vertebra and femur, decreased bone turnover markers (osteocalcin and TNFα) and expression of skeletal osteoclastogenic genes. It also increased new bone formation and expression of osteogenic genes in the femur bone as compared with vehicle groups (Sham) and ovariectomy (OVx), Bortezomib (known PI), injectible parathyroid hormone and alendronate (FDA approved drugs). WFA promoted the process of cortical bone regeneration at drill-holes site in the femur mid-diaphysis region and cortical gap was bridged with woven bone within 11 days of both estrogen sufficient and deficient (ovariectomized, Ovx) mice.
CONCLUSIONS:
Together our data suggest that WFA stimulates bone formation by abrogating proteasomal machinery and provides knowledge base for its clinical evaluation as a bone anabolic agent. |
|
| Cell Research: |
| Vascul Pharmacol. 2014 Mar;60(3):120-6. | | Antiplatelet, anticoagulant, and profibrinolytic activities of withaferin A.[Pubmed: 24534482] | Withaferin A (WFA), an active compound from Withania somnifera, is widely researched for its anti-inflammatory, cardioactive and central nervous system effects. However, antiplatelet, anticoagulant, and profibrinolytic properties of WFA have not been studied.
METHODS AND RESULTS:
In this study, the anticoagulant activities of WFA were measured by monitoring activated partial thromboplastin-time (aPTT), prothrombin time (PT), fibrin polymerization, platelet aggregation, thrombus formation, and the activities of cell-based thrombin and activated factor X (FXa). The effects of WFA on the expressions of plasminogen activator inhibitor type 1 (PAI-1) and tissue-type plasminogen activator (t-PA) were also tested in tumor necrosis factor-α (TNF-α) activated human umbilical vein endothelial cells (HUVECs). Our data showed that WFA inhibited thrombin-catalyzed fibrin polymerization and platelet aggregation, FeCl3-induced thrombus formation, prolonged aPTT and PT significantly and inhibited the activities and production of thrombin and FXa. WFA prolonged in vivo and ex vivo bleeding time and inhibited TNF-α induced PAI-1 production. Furthermore, PAI-1/t-PA ratio was significantly decreased by WFA.
CONCLUSIONS:
Collectively, these results indicate that WFA possesses antithrombotic activities and suggest that the current study could provide bases for the development of new anticoagulant agents. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)