| Description: |
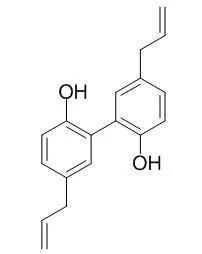
Magnolol is a dual agonist of both RXRα and PPARγ, with EC50 values of 10.4 μM and 17.7 μM, respectively. Magnolol has antifungal, antibacterial, anti-oxidative, antidepressant-like, anti- tumor, and neuroprotective effects, it also prevents skin photoaging in UVB-irradiated hairless mice. Magnolol has a wide spectrum of targets including TNF-α, NF-KB, GSK3β. |
| Targets: |
JNK | ERK | AP-1 | NF-kB | ROS | HO-1 | Nrf2 | MMP(e.g.TIMP) | Antifection | NOS | Akt | GSK-3 | PPARγ | COX | TNF-α | Bcl-2/Bax | cAMP | RXRα |
| In vitro: |
| J Nat Prod. 2015 Jan 23;78(1):61-8. | | Magnolol Inhibits RANKL-induced osteoclast differentiation of raw 264.7 macrophages through heme oxygenase-1-dependent inhibition of NFATc1 expression.[Pubmed: 25574844] | Magnolol (1) isolated from Magnolia officinalis exhibits many beneficial effects such as anti-inflammatory and antioxidant activity. The aim of this study was to evaluate the effects of Magnolol (1) on RANKL-induced osteoclast differentiation and investigate the underlying molecular mechanisms.
METHODS AND RESULTS:
Treatment with Magnolol (1) significantly inhibited osteoclast differentiation of RAW 264.7 macrophages and bone-resorbing activity of osteoclasts in the RANKL-induced system. Moreover, RANKL-activated JNK/ERK/AP-1 and NF-κB signaling, ROS formation, and NFATc1 activation were attenuated by Magnolol (1). A novel finding of this study is that Magnolol (1) can increase heme oxygenase-1 (HO-1) expression and Nrf2 activation in RANKL-stimulated cells. Blocking HO-1 activity with tin protoporphyrin IX markedly reversed Magnolol (1)-mediated inhibition of osteoclast differentiation, NFATc1 nuclear translocation, and MMP-9 activity, suggesting that HO-1 contributes to the attenuation of NFATc1-mediated osteoclastogenesis by Magnolol (1). Therefore, the inhibitory effect of Magnolol (1) on osteoclast differentiation is due to inhibition of MAPK/c-fos/AP-1 and NF-κB signaling as well as ROS production and up-regulation of HO-1 expression, which ultimately suppresses NFATc1 induction.
CONCLUSIONS:
These findings indicate that Magnolol (1) may have potential to treat bone diseases associated with excessive osteoclastogenesis. | | Arch Pharm Res. 2000 Feb;23(1):46-9. | | Antifungal activity of magnolol and honokiol.[Pubmed: 10728656] | Two neolignan compounds, Magnolol (5,5'-diallyl-2,2'-dihydroxybiphenyl, 1) and honokiol (5,5'-diallyl-2,4'-dihydroxybiphenyl, 2), were isolated from the stem bark of Magnolia obovata and evaluated for antifungal activity against various human pathogenic fungi.
METHODS AND RESULTS:
Compound 1 and 2 showed significant inhibitory activities against Trichophyton mentagrophytes, Microsporium gypseum, Epidermophyton floccosum, Aspergillus niger, Cryptococcus neoformans, and Candida albicans with minimum inhibitory concentrations (MIC) in a range of 25-100 microg/ml.
CONCLUSIONS:
Therefore, compound 1 and 2 could be used as lead compounds for the development of novel antifungal agents. | | Eur J Pharmacol. 2004 Aug 2;496(1-3):189-95. | | In vitro antibacterial and anti-inflammatory effects of honokiol and magnolol against Propionibacterium sp.[Pubmed: 15288590] | Honokiol and Magnolol, two major phenolic constituents of Magnolia sp., have been known to exhibit antibacterial activities. However, until now, their antibacterial activity against Propionibacterium sp. has not been reported.
METHODS AND RESULTS:
To this end, the antibacterial activities of honokiol and Magnolol were detected using the disk diffusion method and a two-fold serial dilution assay. Honokiol and Magnolol showed strong antibacterial activities against both Propionibacterium acnes and Propionibacterium granulosum, which are acne-causing bacteria. The minimum inhibitory concentrations (MIC) of honokiol and Magnolol was 3-4 microg/ml (11.3-15 microM) and 9 microg/ml (33.8 microM), respectively. In addition, the killing curve analysis showed that Magnolol and honokiol killed P. acnes rapidly, with 10(5) organisms/ml eliminated within 10 min of treatment with either 45 microg (169.2 microM) of Magnolol or 20 microg (75.2 microM) of honokiol per ml. The cytotoxic effect of honokiol and Magnolol was determined by a colorimetric (3-(4,5-dimetyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide) (MTT) assay using two animal cell lines, human normal fibroblasts and HaCaT. In this experiment, Magnolol exhibited lower cytotoxic effects than honokiol at the same concentration, but they showed similar cytotoxicity when triclosan was employed as an acne-mitigating agent. In addition, they reduced secretion of interleukin-8 and tumor necrosis factor alpha (TNF-alpha) induced by P. acnes in THP-1 cells indicating the anti-inflammatory effects of them. When applied topically, neither phenolic compound induced any adverse reactions in a human skin primary irritation test.
CONCLUSIONS:
Therefore, based on these results, we suggest the possibility that Magnolol and honokiol may be considered as attractive acne-mitigating candidates for topical application. |
|
| In vivo: |
| Toxicol Appl Pharmacol. 2014 Sep 15;279(3):294-302. | | Magnolol protects neurons against ischemia injury via the downregulation of p38/MAPK, CHOP and nitrotyrosine.[Pubmed: 25038313] | Magnolol is isolated from the herb Magnolia officinalis, which has been demonstrated to exert pharmacological effects. Our aim was to investigate whether Magnolol is able to act as an anti-inflammatory agent that brings about neuroprotection using a global ischemic stroke model and to determine the mechanisms involved.
METHODS AND RESULTS:
Rats were treated with and without Magnolol after ischemia reperfusion brain injury by occlusion of the two common carotid arteries. The inflammatory cytokine production in serum and the volume of infarction in the brain were measured. The proteins present in the brains obtained from the stroke animal model (SAM) and control animal groups with and without Magnolol treatment were compared. Magnolol reduces the total infarcted volume by 15% and 30% at dosages of 10 and 30mg/kg, respectively, compared to the untreated SAM group. The levels of acute inflammatory cytokines, including interleukin-1 beta, tumor necrosis factor alpha, and interleukin-6 were attenuated by Magnolol. Magnolol was also able to suppress the production of nitrotyrosine, 4-hydroxy-2-nonenal (4-HNE), inducible NO synthase (iNOS), various phosphorylated p38 mitogen-activated protein kinases and various C/EBP homologues. Furthermore, this modulation of ischemia injury factors in the SAM model group treated with Magnolol seems to result from a suppression of reactive oxygen species production and the upregulation of p-Akt and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB).
CONCLUSIONS:
These findings confirm the anti-oxidative properties of Magnolol, including the inhibition of ischemic injury to neurons; this protective effect seems to involve changes in the in vivo activity of Akt, GSK3β and NF-κB. | | Prog Neuropsychopharmacol Biol Psychiatry. 2008 Apr 1;32(3):715-25. | | Antidepressant-like effects of the mixture of honokiol and magnolol from the barks of Magnolia officinalis in stressed rodents.[Pubmed: 18093712 ] | Honokiol and Magnolol are the main constituents simultaneously identified in the barks of Magnolia officinalis, which have been used in traditional Chinese medicine to treat a variety of mental disorders including depression.
In the present study, we reported on the antidepressant-like effects of oral administration of the mixture of honokiol and Magnolol in well-validated models of depression in rodents: forced swimming test (FST), tail suspension test (TST) and chronic mild stress (CMS) model.
METHODS AND RESULTS:
The mixture of honokiol and Magnolol significantly decreased immobility time in the mouse FST and TST, and reversed CMS-induced reduction in sucrose consumption to prevent anhedonia in rats. However, this mixture was unable to affect ambulatory or rearing behavior in the mouse open-field test. CMS induced alterations in 5-hydroxytryptamine (5-HT) and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) levels in various brain regions of rats. An increase in serum corticosterone concentrations and a reduction in platelet adenylyl cyclase (AC) activity were simultaneously found in the CMS rats. The mixture of honokiol and Magnolol at 20 and 40 mg/kg significantly attenuated CMS-induced decreases of 5-HT levels in frontal cortex, hippocampus, striatum, hypothalamus and nucleus accumbens. And it markedly increased 5-HIAA levels in frontal cortex, striatum and nucleus accumbens at 40 mg/kg and in frontal cortex at 20 mg/kg in the CMS rats. A subsequent reduction in 5-HIAA/5-HT ratio was found in hippocampus and nucleus accumbens in the CMS rats receiving this mixture. Furthermore, the mixture of honokiol and Magnolol reduced elevated corticosterone concentrations in serum to normalize the hypothalamic-pituitary-adrenal (HPA) hyperactivity in the CMS rats. It also reversed CMS-induced reduction in platelet AC activity, via upregulating the cyclic adenosine monophosphate (cAMP) pathway. These results suggested that the mixture of honokiol and Magnolol possessed potent antidepressant-like properties in behaviors involved in normalization of biochemical abnormalities in brain 5-HT and 5-HIAA, serum corticosterone levels and platelet AC activity in the CMS rats.
CONCLUSIONS:
Our findings could provide a basis for examining directly the interaction of the serotonergic system, the HPA axis and AC-cAMP pathway underlying the link between depression and treatment with the mixture of honokiol and Magnolol. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)