| Description: |
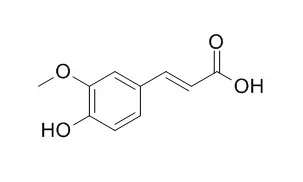
Ferulic acid is an antioxidant found naturally in plant cell walls , shows antioxidant activity in parallel with their radical scavenging activity, it has been approved in certain countries as food additive to prevent lipid peroxidation. Ferulic acid has been reported to have many physiological functions, including antioxidant, antimicrobial, anti-inflammatory, anti-thrombosis, anti- diabetic, and anti-cancer activities; it also protects against coronary disease, lowers cholesterol and increases sperm viability. |
| In vitro: |
| J. Sci. Food Agr., 2004, 84(11):1261-9. | | Ferulic acid: pharmaceutical functions, preparation and applications in foods.[Reference: WebLink] |
Ferulic acid (4‐hydroxy‐3‐methoxycinnamic acid), an effective component of Chinese medicine herbs such as Angelica sinensis, Cimicifuga heracleifolia and Lignsticum chuangxiong, is a ubiquitous phenolic acid in the plant kingdom. It is mainly conjugated with mono‐ and oligosaccharides, polyamines, lipids and polysaccharides and seldom occurs in a free state in plants.
METHODS AND RESULTS:
Ferulic acid is a phenolic acid of low toxicity; it can be absorbed and easily metabolized in the human body. Ferulic acid has been reported to have many physiological functions, including antioxidant, antimicrobial, anti‐inflammatory, anti‐thrombosis, and anti‐cancer activities. It also protects against coronary disease, lowers cholesterol and increases sperm viability. Because of these properties and its low toxicity, Ferulic acid is now widely used in the food and cosmetic industries. It is used as the raw material for the production of vanillin and preservatives, as a cross‐linking agent for the preparation of food gels and edible films, and as an ingredient in sports foods and skin protection agents. Ferulic acid can be prepared by chemical synthesis and through biological transformation.
CONCLUSIONS:
As polysaccharide ferulate is a natural and abundant source of Ferulic acid, preparation of Ferulic acid from plant cell wall materials will be a prospective pathway. | | 2015 Oct 12;16(10):24011-31. | | Ferulic Acid Exerts Anti-Angiogenic and Anti-Tumor Activity by Targeting Fibroblast Growth Factor Receptor 1-Mediated Angiogenesis[Pubmed: 26473837] | | Most anti-angiogenic therapies currently being evaluated target the vascular endothelial growth factor (VEGF) pathway; however, the tumor vasculature can acquire resistance to VEGF-targeted therapy by shifting to other angiogenesis mechanisms. Therefore, other therapeutic agents that block non-VEGF angiogenic pathways need to be evaluated. Here, we identified Ferulic acid as a novel fibroblast growth factor receptor 1 (FGFR1) inhibitor and a novel agent with potential anti-angiogenic and anti-cancer activities. Ferulic acid demonstrated inhibition of endothelial cell proliferation, migration and tube formation in response to basic fibroblast growth factor 1 (FGF1). In ex vivo and in vivo angiogenesis assays, Ferulic acid suppressed FGF1-induced microvessel sprouting of rat aortic rings and angiogenesis. To understand the underlying molecular basis, we examined the effects of Ferulic acid on different molecular components and found that Ferulic acid suppressed FGF1-triggered activation of FGFR1 and phosphatidyl inositol 3-kinase (PI3K)-protein kinase B (Akt) signaling. Moreover, Ferulic acid directly inhibited proliferation and blocked the PI3K-Akt pathway in melanoma cell. In vivo, using a melanoma xenograft model, Ferulic acid showed growth-inhibitory activity associated with inhibition of angiogenesis. Taken together, our results indicate that Ferulic acid targets the FGFR1-mediated PI3K-Akt signaling pathway, leading to the suppression of melanoma growth and angiogenesis.
Keywords: Akt; FGFR1; angiogenesis; Ferulic acid; melanoma. |
|
| In vivo: |
| Phytother Res. 2004 Apr;18(4):310-4. | | Ferulic acid alleviates lipid peroxidation in diabetic rats.[Pubmed: 15162367 ] | Diabetes mellitus is a metabolic disorder associated with increased formation of free radicals. The objective of our study was to determine whether Ferulic acid (FA), a phenolic acid, has any role to play in diabetes induced free radical formation.
METHODS AND RESULTS:
Diabetes was induced with streptozotocin. The levels of blood glucose, thiobarbituric acid reactive substances (TBARS), hydroperoxides and free fatty acids (FFA) increased in the liver of diabetic animals. The activities of glutathione peroxidase (GPx), superoxide dismutase (SOD) and catalase (CAT) decreased in the liver. Histopathology of pancreas also shows shrunken islets. Supplementation of FA to the diabetic rats resulted in a decrease in the levels of glucose, TBARS, hydroperoxides, FFA and an increase in reduced glutathione (GSH). FA also resulted in increased activities of SOD, CAT, GPx and expansion of pancreatic islets. The effect was much pronounced with lower dose treatment.
CONCLUSIONS:
Thus our study shows that administration of Ferulic acid helps in enhancing the antioxidant capacity of these diabetic animals by neutralizing the free radicals formed thereby reducing the intensity of diabetes. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)