| Kinase Assay: |
| J. Korean Soc. Appl. Bi., 2014, 57(4):473-9. | | Pinoresinol Diglucoside is Screened as a Putative alpha-Glucosidase Inhibiting Compound in Actinidia arguta leaves[Reference: WebLink] | Actinidia arguta leaves are consumed as a popular food material in Korea and have been reported to exert beneficial effects on humans due to its constituent polyphenolic compounds.
In this study, the alpha-glucosidase inhibitory compounds in A. arguta were screened and identified through alpha-glucosidase-guided fractionation and metabolomic analysis.
METHODS AND RESULTS:
The 50% ethanol extracts of A. arguta showed strong inhibitory effect (32.6%), which was comparable to acarbose as a positive control (30.0%). Through multiple steps of fractionation, Pinoresinol diglucoside and fertaric acid were identified as the major potent compounds in A. arguta inhibiting alpha-glucosidase activity by liquid chromatography mass spectrometry analysis and metabolomic comparison. Particularly, because pinoresinol and its glycosides have been demonstrated as alpha-glucosidase inhibitory agents, Pinoresinol diglucoside was proposed to be a putative key compound for alpha-glucosidase inhibition in A. arguta.
CONCLUSIONS:
This is the first study demonstrating the anti-diabetic effect of a pinoresinol-containing fraction of A. arguta and would be useful for its application as a natural alpha-glucosidase inhibitor.
|
|
| Structure Identification: |
| Appl Microbiol Biotechnol. 2012 Feb;93(4):1475-83. | | Structure identification and fermentation characteristics of pinoresinol diglucoside produced by Phomopsis sp. isolated from Eucommia ulmoides Oliv.[Pubmed: 22048615] | Pinoresinol diglucoside (PDG) is the important antihypertensive compound in Eucommia ulmoides Oliv., a traditional Chinese herb medicine. The research objective was to certify the possibility of producing PDG through fermentation.
METHODS AND RESULTS:
PDG-producing endophytic fungi were isolated from E. ulmoides Oliv., and the highest PDG-yielding (11.65 mg/L) isolate, XP-8, was identified as Phomopsis sp. according to the morphological characteristics and the phylogenetic tree constructed on the basis of the gene sequence in the internal transcribed spacers district.
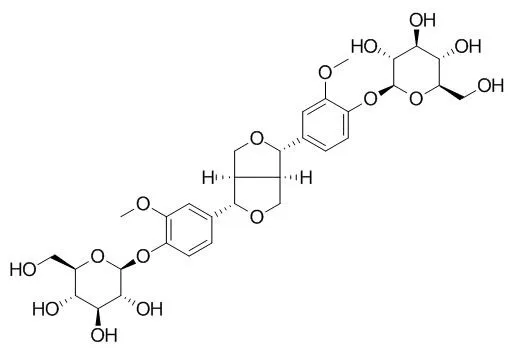
The microbial PDG was isolated by using S-8 resin and purified to a purity of 98.7% using preparative high-performance liquid chromatography (HPLC). Information obtained from the UV spectrum (277 and 227 nm, in water solution), infra-red spectrum (3,428; 2,930; 2,877; 1,637; 1,600; and 1,513; 1,460; 1,421; 1,269; 1,223; 1,075; 658 cm(-1), in powder), molecular weight (682 Da, measured using HPLC-electrospray ionization mass spectrometry (ESI/MS) and tandem mass spectrometry), and nuclear magnetic resonance analysis show the microbial PDG is (+)-1-pinoresinol 4,4'-di-O-β-D-glucopyranoside, same as the plant-derived PDG. The microbial PDG is stable in pH range from 3 to 11 but less stable at temperature higher than 90 °C and in light exposure.
CONCLUSIONS:
During the fermentation, PDG production outside cells starts at the later stage of cell growth when the residual sugar in the medium was low. The study reveals the possibility for production of PDG by fermentation. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)