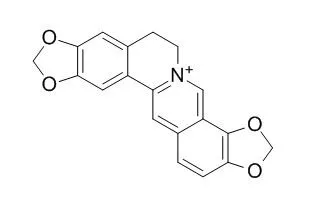
| Description: |
Coptisine is an efficient uncompetitive IDO inhibitor with a Ki value of 5.8 μM and an IC50 value of 6.3 μM, it can consequently prevent neuron loss, reduce amyloid plaque formation, and ameliorate impaired cognition, it could as a potential new class of drugs for AD treatment. Coptisine has cardioprotection, anti- hypercholesterolemia, anti-fungal, anti-osteosarcoma, anti-hepatoma and anti-leukaemia activities, it also has antispasmodic and relaxant activity on a guinea-pig ileum. |
| In vitro: |
| J Appl Microbiol. 2009 Oct;107(4):1072-80. | | Investigation of the anti-fungal activity of coptisine on Candida albicans growth by microcalorimetry combined with principal component analysis.[Pubmed: 19426275] | This study investigated the anti-fungal activity of Coptisine on Candida albicans growth.
METHODS AND RESULTS:
The metabolic power-time curves of Candida albicans growth at 37 degrees C affected by Coptisine were measured by microcalorimetry using an LKB-2277 Bioactivity Monitor with stop-flow mode. Then, the diameter of inhibitory zones in the agar layer was observed using agar cup method, and the minimal inhibitory concentration (MIC) of Coptisine on Candida albicans growth was determined by serial dilution method. From the principal component analysis on nine quantitative parameters obtained from the power-time curves, we could easily evaluate the anti-fungal activity of Coptisine by analysing the change of values of the main two parameters, growth rate constant k and maximum power output in the log phase P(m, log). The results showed that Coptisine had strong anti-fungal activity: at a low concentration (45 microg ml(-1)) began to inhibit the growth of Candida albicans and at a high concentration (500 microg ml(-1)) completely inhibited Candida albicans growth. Coptisine gave big inhibitory zones with diameters between 11 and 43 mm within test range, and the MIC of it was 1000 microg ml(-1).
CONCLUSIONS:
Coptisine had strong anti-fungal activity on Candida albicans growth. The method of microcalorimetry applied for the assay of anti-fungal activity of Coptisine was quantitative, sensitive and simple.
This work will provide useful information for the development of chemical biology policy in the use of anti-microbials in food and drug production. | | Planta Med. 1998 Dec;64(8):758-60. | | Antispasmodic and relaxant activity of chelidonine, protopine, coptisine, and Chelidonium majus extracts on isolated guinea-pig ileum.[Pubmed: 9933996 ] | Two ethanolic dry extracts from the herb Chelidonium majus L. with a defined content of the main alkaloids (chelidonine, protopine, and coptisisine) and the alkaloids themselves were studied in three different antispasmodic test models on isolated ileum of guinea-pigs.
METHODS AND RESULTS:
In the BaCl2-stimulated ileum, chelidonine and protopine exhibited the known papaverine-like musculotropic action, whereas Coptisine (up to 3.0 x 10(-5) g/ml) was ineffective in this model. Both extracts were active with 53.5% and 49.0% relaxation at 5 x 10(-4) g/ml. The carbachol and the electric field stimulated contractions were antagonized by all three alkaloids. Coptisine showed competitive antagonist behaviour with a pA2 value of 5.95. Chelidonine and protopine exhibited a certain degree of non-competitive antagonism. In the electric field the antagonist activities decreased in the order protopine > Coptisine > chelidonine. The concentrations of the chelidonium herb extracts for 50% inhibition of the carbachol and electrical field induced spasms were in the range of 2.5 to 5 x 10(-4) g/ml. | | 2015;43(1):291-302. | | The IDO inhibitor coptisine ameliorates cognitive impairment in a mouse model of Alzheimer's disease[Pubmed: 25079795] | | Indoleamine 2,3-dioxygenase (IDO), the first and rate-limiting enzyme in the kynurenine pathway (KP) of tryptophan catabolism, was recently established as one of the potential players involved in the pathogenesis of Alzheimer's disease (AD). Coptisine is a main pharmacological active constituent of the traditional Chinese medicinal prescription Oren-gedoku-to (OGT) which has therapeutic potential for the treatment of AD. Our recent studies have demonstrated that OGT significantly inhibited recombinant human IDO activity, which shed light on the possible mechanism of OGT's action on AD. Here, we characterized the effects of Coptisine in an AD mouse model on the basis of its IDO inhibitory ability. Coptisine was found to be an efficient uncompetitive IDO inhibitor with a Ki value of 5.8 μM and an IC50 value of 6.3 μM. In AβPP/PS1 transgenic mice, oral administration of Coptisine inhibited IDO in the blood and decreased the activation of microglia and astrocytes, consequently prevented neuron loss, reduced amyloid plaque formation, and ameliorated impaired cognition. Neuronal pheochromocytoma (PC12) cells induced with amyloid-β peptide 1-42 and interferon-γ showed reduction of cell viability and enhancement of IDO activity, while Coptisine treatment increased cell viability based on its reversal effect on the enhanced activity of IDO. In conclusion, our present findings provide further evidence supporting the critical links between IDO, KP, and AD, and demonstrate Coptisine, a novel IDO inhibitor, as a potential new class of drugs for AD treatment.
Keywords: Alzheimer's disease; Coptisine; indoleamine 2, 3-dioxygenase; indoleamine 2, 3-dioxygenase inhibitor; kynurenine pathway. | | 2017 Mar;39(3):1010428317694565. | | Coptisine-induced cell cycle arrest at G2/M phase and reactive oxygen species-dependent mitochondria-mediated apoptosis in non-small-cell lung cancer A549 cells[Pubmed: 28351307] | | This study aimed to explore the effect of Coptisine on non-small-cell lung cancer and its mechanism through various in vitro cellular models (A549). Results claimed significant inhibition of proliferation by Coptisine against A549, H460, and H2170 cells with IC50 values of 18.09, 29.50, and 21.60 μM, respectively. Also, Coptisine exhibited upregulation of pH2AX, cell cycle arrest at G2/M phase, and downregulation of the expression of cyclin B1, cdc2, and cdc25C and upregulation of p21 dose dependently. Furthermore, induction of apoptosis in A549 cells by Coptisine was characterized by the activation of caspase 9, caspase 8, and caspase 3, and cleavage of poly adenosine diphosphate ribose polymerase. In addition, Coptisine was found to increase reactive oxygen species generation, upregulate Bax/Bcl-2 ratio, disrupt mitochondrial membrane potential, and cause cytochrome c release into the cytosol. Besides, treatment with a reactive oxygen species inhibitor (N-acetyl cysteine) abrogated Coptisine-induced growth inhibition, apoptosis, reactive oxygen species generation, and mitochondrial dysfunction. Thus, the mediation of reactive oxygen species in the apoptosis-induced effect of Coptisine in A549 cells was corroborated. These findings have offered new insights into the effect and mechanisms of action of Coptisine against non-small-cell lung cancer.
Keywords: A549 cells; Coptisine; apoptosis; cell cycle arrest; cytotoxicity; reactive oxygen species. |
|
| In vivo: |
| Lipids. 2015 Feb;50(2):185-94. | | The safety and anti-hypercholesterolemic effect of coptisine in Syrian golden hamsters.[Pubmed: 25547428] | Current work was conducted to evaluate the cholesterol-lowering effect of Coptisine extracted from Rhizoma coptidis in Syrian golden hamsters. The safety results indicated that Coptisine was a safe and low-toxic compound.
METHODS AND RESULTS:
Coptisine showed a beneficial effect in the abnormal serum lipid levels induced by a high-fat and high-cholesterol diet (HFHC): at a concentration of 70.05 mg/kg, Coptisine significantly led to a decrease in total cholesterol, triglycerides, and low-density lipoprotein cholesterol (LDL-c) levels by 26.70, 15.38, and 22.22 %, respectively, and high-density lipoprotein cholesterol (HDL-c) was increased by 41.74 % in serum of hamsters (p < 0.01). In addition, total bile acid (TBA) levels in feces of hamsters were elevated after Coptisine administration. Further investigation has suggested that the mRNA and protein expression of 3-hydroxy-3-methyl-glutaryl-CoA reductase (HMGCR) in the liver of hamsters was down-regulated by high-dosage Coptisine treatment (p < 0.05); mRNA and protein expression of low-density lipoprotein receptor (LDLR) and cholesterol 7α-hydroxylase (CYP7A1) were dramatically up-regulated by Coptisine administration. The apical sodium-dependent bile salt transporter expression was down-regulated in the Coptisine-treated animals, but showed no significant differences from the HFHC groups. Taken together, our results demonstrate that a high dosage of Coptisine could inhibit cholesterol synthesis via suppressing the HMGCR expression and promoting the use and excretion of cholesterol via up-regulating LDLR and CYP7A1 expression.
CONCLUSIONS:
These findings suggest a critical role for Coptisine in anti- hypercholesterolemia, and thus it needs to be considered as a potential natural cholesterol lowering agent. | | Toxicol Lett. 2014 May 2;226(3):328-36. | | Unraveling the novel anti-osteosarcoma function of coptisine and its mechanisms.[Pubmed: 24607417] | Uncontrolled cell proliferation and robust angiogenesis play critical roles in osteosarcoma growth and metastasis. In this study we explored novel agents derived from traditional Chinese medicinal herbs that potently inhibit osteosarcoma growth and metastasis.
METHODS AND RESULTS:
Coptisine, an active component of the herb Coptidis rhizoma, markedly inhibited aggressive osteosarcoma cell proliferation. Coptisine induced cell cycle arrest at the G0/G1 phase through downregulation of CDK4 and cyclin D1 expression and effectively suppressed tumor growth in a xenografted mouse model. Coptisine significantly impeded osteosarcoma cell migration, invasion, and capillary-like network formation by decreasing the expression of VE-cadherin and integrin ß3, and diminishing STAT3 phosphorylation. Coptisine significantly elevated blood erythrocyte and hemoglobin levels while still remaining within the normal range. It also moderately increased white blood cell and platelet counts.
CONCLUSIONS:
These data suggest that Coptisine exerts a strong anti-osteosarcoma effect with very low toxicity and is a potential anti-osteosarcoma drug candidate. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)