| In vitro: |
| Food Chem Toxicol. 2014 Feb;64:225-30. | | Discovery of soluble epoxide hydrolase inhibitors from natural products.[Pubmed: 24309146] | With the goal of developing soluble epoxide hydrolase (sEH) inhibitors with novel chemical structures, the sEH inhibitory activities of 30 natural compounds were evaluated using both a fluorescent substrate, 3-phenyl-cyano(6-methoxy-2-naphthalenyl)methyl ester- 2-oxiraneacetic acid, and a physiological substrate, 14,15-epoxyeicosatrienoic acid.
METHODS AND RESULTS:
To evaluate the selectivity of sEH inhibition, the inhibition of microsomal epoxide hydrolase (mEH), which plays a critical role in detoxification of toxic epoxides, was determined using human liver microsomes. Honokiol and β-amyrin acetate, isolated from Magnolia officinalis and Acer mandshuricum, respectively, displayed strong inhibition of sEH activity, with respective IC50 values of 0.57 μM and 3.4 μM determined using the fluorescent substrate, and 1.7 μM and 6.1 μM determined using 14,15-epoxyeicosatrienoic acid. mEH activity was decreased to 49% or 61% of control activity by 25 μM honokiol or β-amyrin acetate, respectively.
CONCLUSIONS:
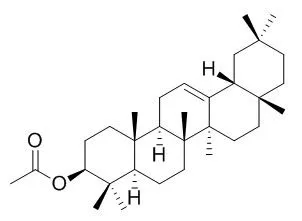
These results suggest that β-amyrin acetate and honokiol exhibit sEH inhibitory activity, although their sEH selectivity should be improved. | | Asian Pac J. Trop. Biomed., 2012, 2(2):S981–S4. | | Antioxidant and Cytotoxicity of β-Amyrin acetate fraction from Bridelia ferruginea Leaves.[Reference: WebLink] | The objective of this work was to determine the beta-Amyrin acetate fraction in leave extract of Bridelia ferruginea and evaluate for its antioxidant and cytotoxicity potentials.
METHODS AND RESULTS:
The dried and pulverized leaves of Bridelia ferruginea was extracted with hexane and then with ethyl acetate. The concentrated ethylacetate extract subjected to silica gel column chromatography and eluted with a mixture of equal volume of hexane and dichloromethane afforded two major fractions. The more polar fraction was concentrated and subjected to GCMS analysis which afforded the steroid, 12-Oleanen-3yl acetate commonly known as beta-Amyrin acetate (66.14%). Its ability to act as a scavenger of DPPH radical and its cytotoxicity potential based on brine shrimp assay were investigated. The DPPH antioxidant assay revealed that the fraction had a higher antioxidant potential with an IC50 value of 158.2μg/mL relative to gallic acid which had IC50 of 201.1 μg/mL. The cytotoxicity assay using the brine shrimp a gave LC50 values of 319 and 5.86 μg/mL for acute and lethal doses respectively indicating extreme toxicity when compared to reference drug, cyclophosphamide which had LC50 value of 2506 μg/mL.
CONCLUSIONS:
Thus, the beta-Amyrin acetate has been identified for the first time in the leave of Bridelia ferruginea. The data here suggest that the beta-Amyrin acetate fraction of the leave of Bridelia ferruginea could be further explored in biological profiling requiring antioxidant and cytotoxic dependent therapeutics as the plant could be a viable source of antioxidant and cytotoxic agents in cancer chemotherapy in the near future. | | Pharm Biol. 2014 Nov;52(11):1478-86. | | beta-Amyrin and alpha-amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity.[Pubmed: 25026352] | To investigate the anti-inflammatory potential of beta-Amyrin acetateand α-amyrin acetate isolated from the stem bark of Alstonia boonei using animal models.
METHODS AND RESULTS:
Chromatographic purification of the crude methanol extract led to the isolation and structure elucidation of beta-Amyrin acetate and α-amyrin acetate.Both beta-Amyrin acetate and α-amyrin acetate inhibited heat-induced hemolysis to as much 47.2 and 61.5%, respectively, while diclofenac sodium (100 μg/mL) evoked only 40.5% inhibition. Both compounds at 100 µg/ear produced significant (p < 0.01) inhibition of ear edema in mice by 39.4 and 55.5%, respectively. Also at 100 mg/kg (p.o.) α-amyrin acetate evoked 60.3% reduction in total leucocyte count and significant (p < 0.05) suppression (47.9%) of neutrophil infiltration.
CONCLUSIONS:
This study generally provided evidence of profound anti-inflammatory activity of beta-Amyrin acetate and α-amyrin acetate isolated from the Alstonia boonei stem bark. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)