| In vitro: |
| Chinese Traditional & Herbal Drugs, 2015 , 46 (11) :1597-602. | | Study on chemical constituents from Re-Du-Ning Injection(Ⅱ)[Reference: WebLink] | To investigate the chemical constituents from Re-Du-Ning Injection(RDN).
METHODS AND RESULTS:
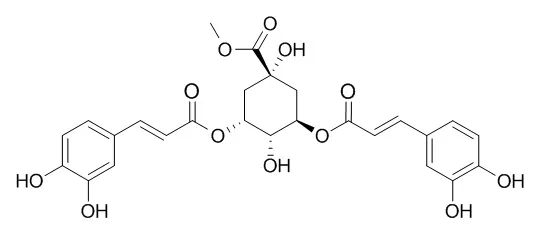
The chemical constituents were isolated by chromatography on silica gel, ODS, Sephadex LH-20, and Toyopearl HW-40 columns and reverse phase MPLC and HPLC repeatedly. Their structures were identified by spectral data and physicochemical property. Sixteen compounds were isolated and identified as 5-O-caffeoylquinic acid(1), 5-O-caffeoylquinic acid methyl ester(2), 4-O-caffeoylquinic acid(3), 5-O-caffeoylquinic acid methyl ester(4), 4,5-di-O-caffeoylquinic acid(5), 4,5-di-O-caffeoylquinic acid methyl ester(6), 3,5-di-O-caffeoylquinic acid(7), 3,5-Di-O-caffeoylquinic acid methyl ester(8), 3,4-di-O-caffeoylquinic acid(9), 3,4-di-O-caffeoylquinic acid methyl ester(10), secologanic acid(11), vogeloside(12), 7-epi-vogeloside(13), E-aldosecologanin(14), Z-aldosecologanin(15), and 5H,8H-pyrano [4,3-d] thiazolo [3,2-a] pyridine-3-carboxylic acid(16). Compounds 1—10 showed high efficiency and low toxicity with antivirus activity against RSV.
CONCLUSIONS:
All the isolated compounds are reported from RDN Injection for the first time, and caffeoylquinic acids may be one of antivirus pharmacodynamic material bases of RDN. | | Arch Pharm Res. 2008 Jul;31(7):900-4. | | Constituents of the flowers of Erigeron annuus with inhibitory activity on the formation of advanced glycation end products (AGEs) and aldose reductase.[Pubmed: 18704333 ] |
METHODS AND RESULTS:
Seven phenolic compounds, caffeic acid (1), 4-hydroxybenzoic acid (2), 4-methoxybenzoic acid (3), protocatechuic acid (4), eugenol O-beta-D: -glucopyranoside (5), 3,6-di-O-feruloylsucrose (6), and 3,5-Di-O-caffeoylquinic acid methyl ester (7), were isolated from an EtOAc-soluble partition of the flowers of Erigeron annuus. The structures of 1-7 were determined by spectroscopic data interpretation, particularly 1D and 2D NMR studies, and by comparison of their data with those published in the literature. All the isolates were subjected to in vitro bioassays to evaluate their inhibitory activities against the formation of advanced glycation end products (AGEs) and rat lens aldose reductase (RLAR).
CONCLUSIONS:
Of the compounds, 1, 6, and 7 exhibited potent inhibitory activities against the formation of AGEs. In the RLAR assay, compound 7 showed the most potent inhibitory activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)