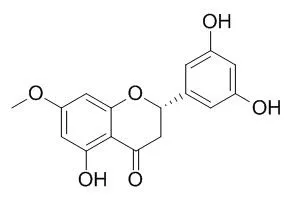
| Structure Identification: |
| Pak J Pharm Sci. 2013 Mar;26(2):375-81. | | Simultaneous quantification of flavonoids in blood plasma by a high-performance liquid chromatography method after oral administration of Blumea balsamifera leaf extracts in rats.[Pubmed: 23455210] | The leaves of Blumea balsamifera are used as a folk medicine in kidney stone diseases in South-East Asia. Phytochemical investigation revealed leaves contained a number of flavonoids.
METHODS AND RESULTS:
In view of these, the present work was aimed to quantify and preliminary pharmacokinetic investigation of five flavonoids viz. dihydroquercetin-7,4¢-dimethyl ether (I), dihydroquercetin-4¢-methyl ether (II), 5,7,3¢,5¢-tetrahydroxyflavanone (III), Blumeatin (IV) and quercetin (V) in rat plasma following oral administration (0.5g/Kg) of B. balsamifera leaf extract in rats. Quantification was achieved by using a validated, reproducible high-performance liquid chromatographic method. The mean recoveries of dihydroquercetin-7,4¢-dimethyl ether (I), dihydroquercetin-4¢-methyl ether (II), 5,7,3¢,5¢-tetrahydroxyflavanone (III), Blumeatin (IV) and quercetin (V) were 90.6, 93.4, 93.5, 91.2 and 90.3% respectively. The limit of quantification was 25 ng/mL for I and IV, 10 ng/mL for II and III and 100 ng/mL for V respectively. The within day and day-to-day precision for all the compounds were < 10%. The validated HPLC method herein was applied for pharmacokinetic studies and the main pharmacokinetic parameters were: t1/2 (hr) 5.8, 4.3, 2.9, 5.7 and 7.3, Cmax (ng/mL) 594.9, 1542.9 1659.9, 208.9 and 3040.4; Tmax (hr) 4.7, 1.0, 1.0, 3.5 and 2.3; AUC0-oo (ng hr/mL) 5040, 5893, 9260, 1064 and 27233 for dihydroquercetin-7,4¢-dimethyl ether (I), dihydroquercetin-4¢-methyl ether (II), 5,7,3¢,5¢-tetrahydroxyflavanone (III), Blumeatin (IV) and quercetin (V) respectively.
CONCLUSIONS:
The developed method was suitable for pharmacokinetic studies and this preliminary study also revealed significant absorption after oral dosing in rats. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)