| Structure Identification: |
| Anal Bioanal Chem. 2014 Apr;406(9-10):2433-44. | | Quantification of cyclic dipeptides from cultures of Lactobacillus brevis R2Δ by HRGC/MS using stable isotope dilution assay.[Pubmed: 24477717 ] | Lactic acid bacteria (LAB) play an important role as natural preservatives in fermented food and beverage systems, reducing the application of chemical additives. Thus, investigating their antifungal compounds, such as cyclic dipeptides, has gained prominence. Previous research has primarily focussed on isolation of these compounds. However, their precise quantification will provide further information regarding their antifungal performance in a complex system.
METHODS AND RESULTS:
To address this, deuterated labelled standards of the cyclic dipeptides cyclo(Leu-Pro), Cyclo(Pro-Pro), cyclo(Met-Pro) and cyclo(Phe-Pro) were synthesized, and stable isotope dilution assays were developed, enabling an accurate quantification of cyclo(Leu-Pro), Cyclo(Pro-Pro), cyclo(Met-Pro) and cyclo(Phe-Pro) in MRS-broth and wort. Quantitative results showed that, in the Lactobacillus brevis R2Δ fermented MRS-broth, the concentrations of cyclo(Leu-Pro), Cyclo(Pro-Pro) and cyclo(Phe-Pro) were significantly higher (P < 0.05), than in wort for cyclo (Leu-Pro) when compared with their controls.
CONCLUSIONS:
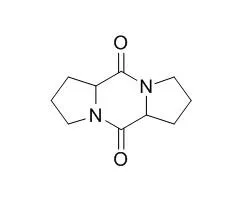
This indicates that the formation of these three cyclic dipeptides is related to L. brevis R2Δ metabolism. Furthermore, this represents the first report of cyclic dipeptides quantification using stable isotope dilution assays in LAB cultures both in vitro and in a food system. | | Org .Lett., 2006, 8(11):2421-4. | | Verpacamides A-D, a sequence of C11N5 diketopiperazines relating cyclo(Pro-Pro) to cyclo(Pro-Arg), from the marine sponge Axinella vaceleti: possible biogenetic precursors of pyrrole-2-aminoimidazole alkaloids.[Pubmed: 16706541 ] | | Four C(11)N(5) diketopiperazine metabolites named verpacamides A (6), B (7), C (8), and D (9) consisting of a proline-arginine dipeptide skeleton have been isolated from the marine sponge Axinella vaceleti. Verpacamides A-D are a sequence of metabolites showing the transformation of proline and arginine into the oxidized guanidinyl-Cyclo(Pro-Pro) 8 and 9. Compounds 6-9 are structurally and chemically related to C(11)N(5) pyrrole-2-aminoimidazole metabolites also isolated from the Axinellidae and Agelasidae families of sponges and exemplified by dispacamide A (4) and dibromophakellin (10). |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)