| Description: |
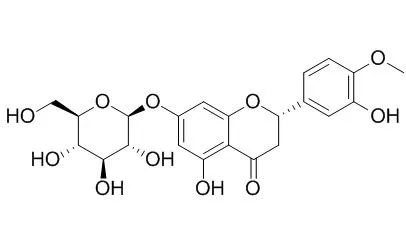
Hesperetin 7-O-glucoside and prunin are direct precursors of naringin and neohesperidin, respectively, in C. aurantium. Hesperetin 7-O-glucoside shows inhibition of human HMG-CoA reductase, it also exhibits effective inhibition of the growth of Helicobacter pylori. Hesperetin 7-O-glucoside can reduce blood pressure in healthy volunteers. |
| Targets: |
HMG-CoA Reductase | Antifection |
| In vitro: |
| Food Chem. 2012 Dec 15;135(4):2253-9. | | Enzymatic bioconversion of citrus hesperidin by Aspergillus sojae naringinase: enhanced solubility of hesperetin-7-O-glucoside with in vitro inhibition of human intestinal maltase, HMG-CoA reductase, and growth of Helicobacter pylori.[Pubmed: 22980799] |
METHODS AND RESULTS:
Hesperetin 7-O-glucoside (Hes-7-G) was produced by the enzymatic conversion of hesperidin by Aspergillus sojae naringinase due to the removal of the terminal rhamnose.Extracts from orange juice and peel containing the hesperidin were so treated by this enzyme that the hesperidin could also be converted to Hesperetin 7-O-glucoside . The solubility of Hesperetin 7-O-glucoside in 10% ethanol was enhanced 55- and 88-fold over those of hesperidin and hesperetin, respectively, which may make Hesperetin 7-O-glucoside more bioavailable. Hesperetin 7-O-glucoside was 1.7- and 2.4-fold better than hesperidin and hesperetin, respectively, in the inhibition of human intestinal maltase. Hesperetin 7-O-glucoside was more potent by 2- and 4-fold than hesperidin in the inhibition of human HMG-CoA reductase. Additionally, Hesperetin 7-O-glucoside exhibited more effective inhibition of the growth of Helicobacter pylori than hesperetin, while its effectiveness was similar to that of hesperidin.
CONCLUSIONS:
Therefore, the results suggest that bioconverted Hesperetin 7-O-glucoside is more effective and bioavailable than hesperidin, as it has enhanced inhibitory and solubility properties. |
|
| In vivo: |
| Mol Nutr Food Res. 2015 May 27. | | Gastrointestinal absorption and metabolism of hesperetin-7-O-rutinoside and hesperetin-7-O-glucoside in healthy humans.[Pubmed: 26018925] | Hesperetin 7-O-glucoside (hesperidin) reduces blood pressure in healthy volunteers but its intestinal absorption and metabolism are not fully understood. Therefore, we aimed to determine sites of absorption and metabolism of dietary flavanone glycosides in humans.
METHODS AND RESULTS:
Using a single blind, randomized crossover design, we perfused equimolar amounts of Hesperetin 7-O-glucoside and Hesperetin 7-O-glucoside directly into the proximal jejunum of healthy volunteers. We assessed the appearance of metabolites in the perfusate, blood and urine, to determine the sites of metabolism and excretion, and compared this to oral administration. Theglucoside was rapidly hydrolysed by brush border enzymes without any contribution from pancreatic, stomach or other secreted enzymes, or from bacterial enzymes. Only ∼3% of the dose was recovered intact in the perfusate, indicating high absorption. A proportion was effluxed directly back into the perfused segment mainly in the form of hesperetin-3'-O-sulfate. In contrast, very little hydrolysis or absorption of Hesperetin 7-O-glucoside was observed with ∼80% recovered in the perfusate, no hesperetin metabolites were detected in blood and only traces were excreted in urine.
CONCLUSIONS:
The data elucidate the pathways of metabolism of dietary hesperidin in vivo and will facilitate better design of mechanistic studies both in vivo and in vitro. The trial was registered at Medical Faculty of the University of Tübingen. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)