| Description: |
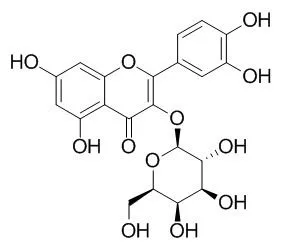
Hyperoside, a naturally occuring flavonoid compound, exerts multiple bioactivities, including myocardial protection, anti-redox, neuroprotective, antifungal, hepatoprotective, anti-inflammatory and antioxidative effects. Hyperoside is a potent natural activator of Nur77 receptor, and a potent selective CYP2D6 inhibitor; it inhibited the HMGB1 signaling pathway, and inhibited the PI3K/Akt/Bad/Bcl XL -regulated mitochondrial apoptotic pathway. Hyperoside is a strong inhibitor of HBsAg and HBeAgsecretion in 2.2.15 cells and DHBV-DNA levels in the HBV-infected duck model. |
| In vitro: |
| Int J Mol Sci. 2013 Nov 18;14(11):22697-707. | | Hyperoside downregulates the receptor for advanced glycation end products (RAGE) and promotes proliferation in ECV304 cells via the c-Jun N-terminal kinases (JNK) pathway following stimulation by advanced glycation end-products in vitro.[Pubmed: 24252909] | Hyperoside is a major active constituent in many medicinal plants which are traditionally used in Chinese medicines for their neuroprotective, anti-inflammatory and antioxidative effects.
The molecular mechanisms underlying these effects are unknown.
METHODS AND RESULTS:
In this study, quiescent ECV304 cells were treated in vitro with advanced glycation end products (AGEs) in the presence or absence of Hyperoside. The results demonstrated that AGEs induced c-Jun N-terminal kinases (JNK) activation and apoptosis in ECV304 cells. Hyperoside inhibited these effects and promoted ECV304 cell proliferation. Furthermore, Hyperoside significantly inhibited RAGE expression in AGE-stimulated ECV304 cells, whereas knockdown of RAGE inhibited AGE-induced JNK activation. These results suggested that AGEs may promote JNK activation, leading to viability inhibition of ECV304 cells via the RAGE signaling pathway. These effects could be inhibited by Hyperoside.
CONCLUSIONS:
Our findings suggest a novel role for Hyperoside in the treatment and prevention of diabetes. | | J Agric Food Chem. 2005 Jan 12;53(1):32-7. | | Antifungal activity of camptothecin, trifolin, and hyperoside isolated from Camptotheca acuminata.[Pubmed: 15631505] | Leaf spots and root rots are major fungal diseases in Camptotheca acuminata that limit cultivation of the plant for camptothecin (CPT), a promising anticancer and antiviral alkaloid.
METHODS AND RESULTS:
Bioassays showed that pure CPT and flavonoids (trifolin and Hyperoside) isolated from Camptotheca effectively control fungal pathogens in vitro, including Alternaria alternata, Epicoccum nigrum, Pestalotia guepinii, Drechslera sp., and Fusarium avenaceum, although antifungal activity of these compounds in the plant is limited. CPT inhibited mycelial growth by approximately 50% (EC50) at 10-30 microg/mL and fully inhibited growth at 75-125 microg/mL.
The flavonoids were less effective than CPT at 50 microg/mL, particularly within 20 days after treatment, but more effective at 100 or 150 microg/mL.
CONCLUSIONS:
CPT, trifolin, and Hyperoside may serve as leads for the development of fungicides. | | Eur J Pharmacol. 2011 Dec 15;672(1-3):45-55. | | Hyperoside protects primary rat cortical neurons from neurotoxicity induced by amyloid β-protein via the PI3K/Akt/Bad/Bcl(XL)-regulated mitochondrial apoptotic pathway.[Pubmed: 21978835 ] | Amyloid β-protein (Aβ), which is deposited in neurons as neurofibrillary tangles, is known to exert cytotoxic effects by inducing mitochondrial dysfunction. Additionally, the PI3K/Akt-mediated interaction between Bad and Bcl(XL) plays an important role in maintaining mitochondrial integrity. However, the application of therapeutic drugs, especially natural products in Alzheimer's disease therapy via PI3K/Akt/Bad/Bcl(XL)-regulated mitochondrial apoptotic pathway has not aroused extensive attention.
METHODS AND RESULTS:
In the present study, we investigated the neuroprotective effects of Hyperoside, a bioactive flavonoid compound from Hypericum perforatum, on Aβ(25-35)-induced primary cultured cortical neurons, and also examined the potential cellular signaling mechanism for Aβ detoxication. Our results showed that treatment with Hyperoside significantly inhibited Aβ(25-35)-induced cytotoxicity and apoptosis by reversing Aβ-induced mitochondrial dysfunction, including mitochondrial membrane potential decrease, reactive oxygen species production, and mitochondrial release of cytochrome c. Further study indicated that Hyperoside can activate the PI3K/Akt signaling pathway, resulting in inhibition of the interaction between Bad and Bcl(XL), without effects on the interaction between Bad and Bcl-2. Furthermore, Hyperoside inhibited mitochondria-dependent downstream caspase-mediated apoptotic pathway, such as that involving caspase-9, caspase-3, and poly ADP-ribose polymerase (PARP).
CONCLUSIONS:
These results demonstrate that Hyperoside can protect Aβ-induced primary cultured cortical neurons via PI3K/Akt/Bad/Bcl(XL)-regulated mitochondrial apoptotic pathway, and they raise the possibility that Hyperoside could be developed into a clinically valuable treatment for Alzheimer's disease and other neuronal degenerative diseases associated with mitochondrial dysfunction. |
|
| In vivo: |
| Inflammation. 2015 Apr;38(2):784-99. | | Anti-inflammatory effects of hyperoside in human endothelial cells and in mice.[Pubmed: 25097077] |
High-mobility group box 1 (HMGB1) was recently shown to be an important extracellular mediator of systemic inflammation, and endothelial cell protein C receptor (EPCR) has been shown to be involved in vascular inflammation. Hyperoside is an active compound isolated from Rhododendron brachycarpum G. Don (Ericaceae) that was reported to have anti-oxidant, anti-hyperglycemic, anti-cancer, and anti-coagulant activities.
METHODS AND RESULTS:
Here, we show, for the first time, the anti-septic effects of Hyperoside in HMGB1-mediated inflammatory responses and on the shedding of EPCR in vitro and in vivo. The data showed that Hyperoside posttreatment suppressed lipopolysaccharide (LPS)-mediated release of HMGB1 and HMGB1-mediated cytoskeletal rearrangement. Hyperoside also inhibited HMGB1-mediated hyperpermeability and leukocyte migration in septic mice and phorbol-12-myristate 13-acetate (PMA) of cecal ligation and puncture (CLP)-induced EPCR shedding. In addition, Hyperoside inhibited the production of tumor necrosis factor-α (TNF-α) and the HMGB1-mediated activation of Akt, nuclear factor-κB (NF-κB), and extracellular regulated kinase (ERK) 1/2 in HUVECs. Hyperoside also reduced the CLP-induced release of HMGB1, the production of interleukin (IL)-1β, and septic mortality.
CONCLUSIONS:
Collectively, these results suggest Hyperoside as a candidate therapeutic agent for the treatment of vascular inflammatory diseases via inhibition of the HMGB1 signaling pathway. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)