| Kinase Assay: |
| Bioorg Chem. 2017 Feb;70:192-198. | | 8-Hydroxyirilone 5-methyl ether and 8-hydroxyirilone, new antioxidant and α-amylase inhibitors isoflavonoids from Iris germanica rhizomes.[Pubmed: 28069265 ] | Iris species are well recognized as wealthy sources of isoflavonoids.
METHODS AND RESULTS:
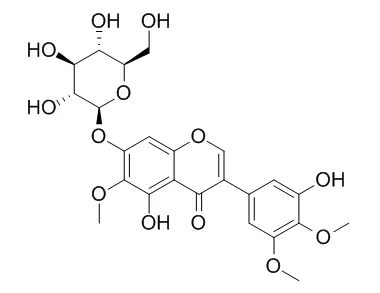
In the present study, phytochemical investigation of the rhizomes of Iris germanica (Iridaceae) procure the isolation of two new isoflavonoids namely, 8-hydroxyirilone 5-methyl ether (2) and 8-hydroxyirilone (3), along with eight known isoflavonoids: irilone 4'-methyl ether (1), irilone (4), irisolidone (5), irigenin S (6), irigenin (7), irilone 4'-O-β-d-glucopyranoside (8), Iridin S (9), and Iridin (10). The isolated flavonoids were structurally characterized with the assist of comprehensive spectroscopic analyses (UV, IR, 1D and 2D NMR, and HRMS) and comparing with the published data. They were estimated for their antioxidant and antidaibetic capacities using DPPH and α-amylase inhibition assays, respectively.
CONCLUSIONS:
Compounds 2, 3, and 4 exhibited prominent antioxidant activities with IC50 values of 12.92, 9.23, and 10.46μM, respectively compared to propyl gallate (IC50 7.11μM). Moreover, 2-5 possessed highest α-amylase inhibitory activity with % inhibition 66.1, 78.3, 67.3, and 70.1, respectively in comparison to acarbose (reference α-amylase inhibitor). Additionally, their structure-activity relationship has been discussed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)