Lemons are a widely used citrus crop and have shown several potential health benefits. In the present study, the mechanism and effectiveness of the anti-cancer and anti-aromatase properties of limonoids were investigated for the first time. Defatted lemon (Citrus lemon L. Burm) seed powder was extracted with ethyl acetate (EtOAc) and methanol (MeOH) for 16 h each, successively.
METHODS AND RESULTS:
These extracts were fractionated using 1D (silica) and 2D (ion exchange and SP-70 columns) column chromatography to obtain nine limonoids.
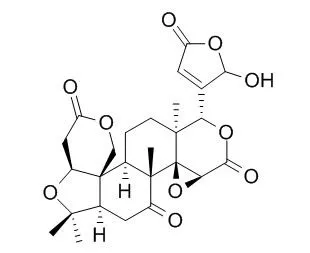
The compounds were identified by TLC, HPLC, and LC-MS techniques. A panel of 9 purified limonoids, including limonin, nomilin, obacunone, Limonexic acid (LNA), isoLimonexic acid (ILNA), nomilinic acid glucoside (NAG), deacetyl nomilinic acid glucoside (DNAG), limonin glucoside (LG) and obacunone glucoside (OG) as well as 4 modified compounds such as limonin methoxime (LM), limonin oxime (LO), defuran limonin (DL), and defuran nomilin (DN), were screened for their cytotoxicity on estrogen receptor (ER)-positive (MCF-7) or ER-negative (MDA-MB-231) human breast cancer cells. We further tested the mechanism of the anti-proliferative activity of limonoids using an in vitro aromatase enzyme assay and western blot with anti-caspase-7. Among the tested limonoids, 11 limonoids exhibited cytotoxicity on MCF-7 whereas 8 limonoids showed cytotoxicity against the MDA-MB-231 cell lines. Although most of the limonoids showed anti-aromatase activity, the inhibition of proliferation was not related to the anti-aromatase activity. On the other hand, the anti-proliferative activity was significantly correlated with caspase-7 activation by limonoids.
CONCLUSIONS:
Our findings indicated that the citrus limonoids may have potential for the prevention of estrogen-responsive breast cancer (MCF-7) via caspase-7 dependent pathways. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)