| Description: |
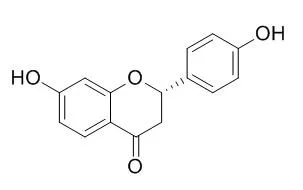
Liquiritigenin, is an estrogenic compound which acts as an agonist selective for the β-subtype of the estrogen receptor, possesses anti-inflammatory, antihyperlipidemic, and antiallergic effects. Liquiritigenin can dose dependently alleviate mechanical, thermal and cold hyperalgesia, it may be potentially useful novel treatments for neuropathic pain. It exhibits antitumour action in pituitary adenoma cells via Ras/ERKs and ROS-dependent mitochondrial signalling pathways. |
| Targets: |
GSK-3 | TNF-α | IL Receptor | IkB | p65 | ROS | JNK | Caspase | Bcl-2/Bax | p38MAPK | ERK | MMP(e.g.TIMP) | IKK |
| In vitro: |
| Biomed Res Int. 2014;2014:965316. | | Liquiritigenin induces tumor cell death through mitogen-activated protein kinase- (MPAKs-) mediated pathway in hepatocellular carcinoma cells.[Pubmed: 24738081] | Liquiritigenin (LQ), separated from Glycyrrhiza radix, possesses anti-inflammatory, antihyperlipidemic, and antiallergic effects.
METHODS AND RESULTS:
Our present study aims to investigate the antihepatocellular carcinoma effects of LQ both in cell and animal models. LQ strikingly reduced cell viability, enhanced apoptotic rate, induced lactate dehydrogenase over-release, and increased intracellular reactive oxygen species (ROS) level and caspase 3 activity in both PLC/PRL/5 and HepG2 cells. The expression of cleaved PARP, the hall-marker of apoptosis, was enhanced by LQ. LQ treatment resulted in a reduction of the expressions of B-cell lymphoma 2 (Bcl-2) and B-cell lymphoma-extra large (Bcl-xL), and an increase of the phosphorylation of c-Jun N-terminal kinases (JNK) and P38. LQ-mediated cell viability reduction, mitochondrial dysfunction, apoptosis related protein abnormal expressions, and JNK and P38 activation were partially abolished by N-Acetyl-L-cysteine (a ROS inhibitor) pretreatment. Moreover, LQ suppressed the activation of extracellular signaling-regulated kinase (ERKs) and reduced the translocation of phosphor-ERKs from cytoplasm to nucleus. This antitumor activity was further confirmed in PLC/PRL/5-xenografted mice model. All these data indicate that the antihepatocellular carcinoma effects of LQ are related to its modulation of the activations of mitogen-activated protein kinase (MAPKs).
CONCLUSIONS:
The study provides experimental evidence supporting LQ as a potential therapeutic agent for hepatocellular carcinoma treatment. | | 2014 Jul 25;5(7):1432-40. | | Protective effect of liquiritigenin against methylglyoxal cytotoxicity in osteoblastic MC3T3-E1 cells[Pubmed: 24789098] | | Methylglyoxal (MG), a reactive dicarbonyl compound, is a metabolic byproduct of glycolysis and elevated MG levels contribute to diabetic complications. Glycation reactions of MG with amino acids can induce oxidative stress, leading to subsequent cytotoxicity. In the present study, the effect of Liquiritigenin on MG-induced cytotoxicity was investigated using osteoblastic MC3T3-E1 cells. Pretreatment of MC3T3-E1 cells with Liquiritigenin prevented the MG-induced cell death and production of protein adduct, intracellular reactive oxygen species, mitochondrial superoxide, cardiolipin peroxidation, and TNF-α in osteoblastic MC3T3-E1 cells. In addition, Liquiritigenin increased the activity of glyoxalase I inhibited by MG. These findings suggest that Liquiritigenin provides a protective action against MG-induced cell damage by reducing oxidative stress and by increasing MG detoxification. Pretreatment with Liquiritigenin prior to MG exposure reduced MG-induced mitochondrial dysfunction by preventing mitochondrial membrane potential dissipation and adenosine triphosphate loss. Additionally, the nitric oxide and PGC-1α levels were significantly increased by Liquiritigenin, suggesting that Liquiritigenin may induce mitochondrial biogenesis. Our findings indicate that Liquiritigenin might exert its therapeutic effects via enhancement of glyoxalase I activity and mitochondrial function, and anti-oxidant and anti-inflammatory activities. Taken together, Liquiritigenin has potential as a preventive agent against the development of diabetic osteopathy related to MG-induced oxidative stress in diabetes. |
|
| In vivo: |
| J Gastroenterol Hepatol. 2014 Oct 14. | | Liquiritigenin ameliorates TNBS-induced colitis in mice.[Pubmed: 25311527] | The anti-inflammatory effects of Liquiritigenin, a major flavonoid isolated from Glycyrrhizae uralensis, have been reported in many inflammation models. However, its protective effects have not been reported in a colitis model. This study investigated the anti-inflammatory effect and mechanism of Liquiritigenin for trinitrobenzene sulfonic acid (TNBS)-induced colitis in mice.
METHODS AND RESULTS:
Male mice imprinting control regions (ICR) were randomly divided into five groups: normal, TNBS-induced colitis, colitis treated with Liquiritigenin at low dose (10 mg/kg) and high dose (20 mg/kg), or mesalazine (10 mg/kg). TNBS colitis induction was performed except for in the normal group, and they were treated with Liquiritigenin or mesalazine except control group. The treatment effect was measured after three days treatment, by body weight, colon length, macroscopic score, histological score, levels of cytokines (tumor necrosis factor-α, interleukin [IL]-1β, IL-6, and IL-10) in colon tissue as well as the nuclear factor kappa-light-chain-enhancer pathway of activated B cells (NF-κB) activation.
Mice treated with high-dose Liquiritigenin showed significant body weight gain, inhibition of colon shortening, protective effect on histological damages, and myeloperoxidase activity of colon tissue compared with the control group. Furthermore, mice treated with high-dose Liquiritigenin experienced significantly suppressed tumor necrosis factor-α, IL-1β, and IL-6 as well as enhanced IL-10 expression (all P < 0.05). High-dose Liquiritigenin treatment group showed significant decreases in TNBS-induced phosphorylation of IKKβ, p65, and IκB-α.
CONCLUSIONS:
Liquiritigenin may ameliorate TNBS-induced colitis in mice by suppressing expression of pro-inflammatory cytokines through NF-κB pathway. | | Sci Rep. 2014 Jul 14;4:5676. | | Liquiritigenin alleviates mechanical and cold hyperalgesia in a rat neuropathic pain model.[Pubmed: 25022218] | This study assessed the potential antinociceptive effects of Liquiritigenin, a plant-derived compound with transient receptor potential melastatin 3 blocking activity in a rat model of persistent neuropathic pain.
METHODS AND RESULTS:
Chronic constriction injury (CCI) to the sciatic nerve was induced in male Sprague-Dawley rats to model human peripheral neuropathic pain. Liquiritigenin (1, 3, or 9 mg/kg) was administered intraperitoneally to examine the effects on mechanical, thermal, and cold hyperalgesia using the von Frey test, plantar test, and cold plate test, respectively. A rotarod test was also conducted to examine motor function. Liquiritigenin dose dependently alleviated mechanical, thermal and cold hyperalgesia. In addition, daily repeated treatment with Liquiritigenin did not demonstrate significant antinociceptive tolerance in the measures of hyperalgesia. Within the doses studied, Liquiritigenin did not significantly affect motor performance.
CONCLUSIONS:
These results suggest that Liquiritigenin may be potentially useful novel treatments for neuropathic pain. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)