| In vitro: |
| Phytochemistry. 2015 Feb;110:133-9. | | Lanostanoids with acetylcholinesterase inhibitory activity from the mushroom Haddowia longipes.[Pubmed: 25577284 ] |
METHODS AND RESULTS:
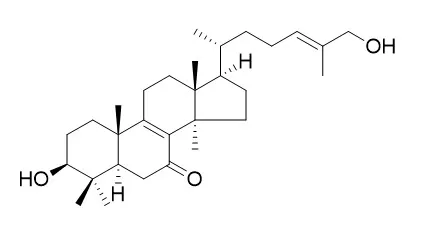
Nine lanostanoids, together with nine known ones, were isolated from the ethyl acetate extract of the fruiting bodies of the mushroom Haddowia longipes. Their structures were elucidated as 11-oxo-ganoderiol D, lanosta-8-en-7,11-dioxo-3β-acetyloxy-24,25,26-trihydroxy, lanosta-8-en-7-oxo-3β-acetyloxy-11β,24,25,26-tetrahydroxy, lanosta-7,9(11)-dien-3β-acetyloxy-24,25,26-trihydroxy, lanosta-7,9(11)-dien-3β-acetyloxy-24,26-dihydroxy-25-methoxy, 11-oxo-Lucidadiol, 11β-hydroxy-Lucidadiol, lucidone H and lanosta-7,9(11),24E-trien-3β-acetyloxy-26,27-dihydroxy by analysing their 1D/2D NMR and MS spectra.
CONCLUSIONS:
In addition, bioassays of inhibitory activity against acetylcholinesterase (AChE) of all compounds showed that thirteen compounds possessed inhibitory activity against AChE with the percentage inhibition ranging from 10.3% to 42.1% when tested at 100 μM. | | Bioorg Med Chem Lett. 2011 Nov 1;21(21):6603-7. | | Selective cholinesterase inhibition by lanostane triterpenes from fruiting bodies of Ganoderma lucidum.[Pubmed: 21924611 ] |
METHODS AND RESULTS:
Two new lanostane triterpenes, named methyl ganoderate A acetonide (1) and n-butyl ganoderate H (2), were isolated from the fruiting bodies of Ganoderma lucidum together with 16 known compounds (3-18). Extensive spectroscopic and chemical studies established the structures of these compounds as methyl 7β,15α-isopropylidenedioxy-3,11,23-trioxo-5α-lanost-8-en-26-oate (1) and n-butyl 12β-acetoxy-3β-hydroxy-7,11,15,23-tetraoxo-5α-lanost-8-en-26-oate (2). Because new compounds exhibiting specific anti-acetylcholinesterase activity are being sought as possible drug candidates for the treatment of Alzheimer's and related neurodegenerative diseases, compounds 1-18 were examined for their inhibitory activities against acetylcholinesterase and butyrylcholinesterase. All of the compounds exhibited moderate acetylcholinesterase-inhibitory activity, with IC(50) values ranging from 9.40 to 31.03μM. In contrast, none of the compounds except Lucidadiol (13) and lucidenic acid N (14) exhibited butyrylcholinesterase-inhibitory activity at concentrations up to 200μM.
CONCLUSIONS:
These results indicate that these lanostane triterpenes are preferential inhibitors of acetylcholinesterase and may be suitable drug candidates. | | Fitoterapia. 2003 Feb;74(1-2):177-80. | | Antiviral lanostanoid triterpenes from the fungus Ganoderma pfeifferi.[Pubmed: 12628419] | | Ganodermadiol, Lucidadiol and applanoxidic acid G were isolated as first triterpenes from the European Basidiomycete Ganoderma pfeifferi. The compounds show antiviral activity against influenza virus type A and HSV type 1. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)