| In vitro: |
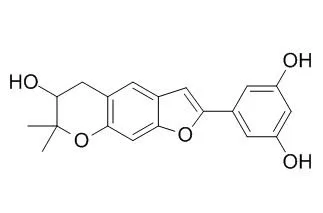
| Arch Pharm Res. 2011 Aug;34(8):1373-80. | | Inhibitory effect of 2-arylbenzofurans from the Mori Cortex Radicis (Moraceae) on oxygen glucose deprivation (OGD)-induced cell death of SH-SY5Y cells.[Pubmed: 21910060] | Three known 2-arylbenzofurans, Moracin P (1), moracin O (2) and mulberrofuran Q (3) were isolated from the MeOH extract of the Mori Cortex Radicis.
METHODS AND RESULTS:
These compounds 1-3 enhanced cell viability in dose-dependent manner against oxygen-glucose deprivation (OGD)-induced cell death in neuroblastoma SH-SY5Y cells, which was measured by MTT reduction assay. (EC(50) values of 10.4, 12.6, and 15.9 μM, respectively). In addition, the compounds 1-3 were examined for their inhibitory effect on OGD-induced ROS production by FACS analysis. We observed these compounds reduced ROS production in OGD-induced cell death (IC(50) values of 1.9, 0.3 and 12.1 μM, respectively). Consequently, reactive oxygen species (ROS) were overexpressed in OGD-induced cells and all three compounds reduced ROS induced by OGD in dosedependent manner.
CONCLUSIONS:
Taken together, compounds 1-3 might protect neuronal cell death against the oxidative stress induced by OGD, though further studies in vitro and in vivo models are necessary. | | Molecules. 2011 Jul 19;16(7):6010-22. | | Inhibitory effects of constituents from Morus alba var. multicaulis on differentiation of 3T3-L1 cells and nitric oxide production in RAW264.7 cells.[Pubmed: 21772233] | A new arylbenzofuran, 3',5'-dihydroxy-6-methoxy-7-prenyl-2-arylbenzofuran (1), and 25 known compounds, including moracin R (2), moracin C (3), moracin O (4), Moracin P (5), artoindonesianin O (6), moracin D (7), alabafuran A (8), mulberrofuran L (9), mulberrofuran Y (10), kuwanon A (11), kuwanon C (12), kuwanon T (13), morusin (14), kuwanon E (15), sanggenon F (16), betulinic acid (17), uvaol (18), ursolic acid (19), β-sitosterol (20), oxyresveratrol 2-O-β-D-glucopyranoside (21), mulberroside A (22), mulberroside B (23), 5,7-dihydroxycoumarin 7-O-β-D-glucopyranoside (24), 5,7-dihydroxycoumarin 7-O-β-D-apiofuranosyl-(1→6)-O-β-D-glucopyranoside (25) and adenosine (26), were isolated from Morus alba var. multicaulis Perro. (Moraceae).
METHODS AND RESULTS:
Their structures were determined by spectroscopic methods. The prenyl-flavonoids 11-14, 16, triterpenoids 17,18 and 20 showed significant inhibitory activity towards the differentiation of 3T3-L1 adipocytes.
CONCLUSIONS:
The arylbenzofurans 1-10 and prenyl-flavonoids 11-16 also showed significant nitric oxide (NO) production inhibitory effects in RAW264.7 cells. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)