| In vitro: |
| Phytomedicine. 2001 Nov;8(6):489-91. | | An antiproliferative norditerpene dilactone, Nagilactone C, from Podocarpus neriifolius.[Pubmed: 11824527] |
METHODS AND RESULTS:
An ethanolic extract of Podocarpus neriifolius D. Don (Podocarpaceae) showed antiproliferative activity against two major tumor cell lines, viz. human HT-1080 fibrosarcoma and murine color 26-L5 carcinoma. Bioassay guided fractionation showed the highest antiproliferative activity in chloroform-soluble fraction. Nagilactone C, the major constituent of this fraction was isolated and characterized by using NMR, IR and FAB-MS spectroscopic methods.
CONCLUSIONS:
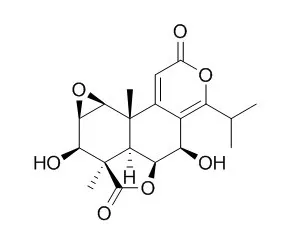
Nagilactone C possessed potent antiproliferative activity against human fibrosarcoma and murine colon carcinoma tumor cell lines exhibiting ED50 values of 2.3 and 1.2 microg/ml, respectively. Hence, Nagilactone C could be the active constituent present in this plant. | | J Chem Ecol. 2001 Jul;27(7):1345-53. | | Sequestration of host plant-derived compounds by geometrid moth, Milionia basalis, toxic to a predatory stink bug, Eocanthecona furcellata.[Pubmed: 11504032] | A predatory stink bug, Eocanthecona furcellata, died after feeding on Milionia basalis larvae.
METHODS AND RESULTS:
The compounds toxic to E. furcellata were isolated from the hemolymph of M. basalis larvae and identified as inumakilactone A, Nagilactone C, and Nagilactone C glucoside. The concentrations of inumakilactone A, Nagilactone C, and Nagilactone C glucoside in the hemolymph of the final instar larvae were 130, 50, and 770 microg/ml, respectively. Nagilactone C showed the highest insecticidal activity against second-instar nymphs of E. furcellata, while Nagilactone C glucoside showed the lowest, one twentieth of that of Nagilactone C. When mixed compounds were given at the same concentrations as those in hemolymph of M. basalis, all nymphs of E. furcellata died with in three days. Inumakilactone A and Nagilactone C were found to be in the leaves of podocarp, Podocarpus macrophyllus, the only host plant of M. basalis, at concentrations of 13 and 175 microg/g fresh weight, respectively. However, no Nagilactone C glucoside was detected in the leaves of this species.
CONCLUSIONS:
These results suggested that M. basalis may transform Nagilactone C to its glucoside. | | Chem Pharm Bull (Tokyo). 2008 Apr;56(4):585-8. | | Cytotoxic constituents from Podocarpus fasciculus.[Pubmed: 18379113] |
METHODS AND RESULTS:
A new diterpene, 16-hydroxy communic acid (1), along with thirty one known compounds including five norditerpenes (2-6), twenty two flavonoids containing four biflavonoids (7-10), nine monoflavonoids (11-19) and nine flavanoid glycosides (20-28), as well as four phenolic constituents (29-32) were isolated from the 95% ethanolic extract of Podocarpus fasciculus. The structure of 1 was elucidated using spectral methods.
CONCLUSIONS:
Of these isolates, Nagilactone C (2) showed the most significant inhibitory effects against DLD cells (human colon carcinoma) (ED(50)=2.57 microg/ml) and compounds 7, 8, 10, 11, and 12 had moderate cytotoxic activity against human KB (human oral epithelium carcinoma), Hela (human cervical carcinoma), Hepa (human hepatoma), DLD (colon carcinoma), and A-549 (human lung carcinoma) tumor cell lines.Preliminary structure-activity relationship studies of the isolated diterpenoids and biflavonoids are discussed. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)