| In vitro: |
| Nat Prod Res. 2013;27(17):1521-7. | | Isolation of the volatile fraction from Apium graveolens L. (Apiaceae) by supercritical carbon dioxide extraction and hydrodistillation: chemical composition and antifungal activity.[Pubmed: 22974401 ] | Apium graveolens L. (wild celery), belonging to the family of Apiaceae, is a scaposus hemicryptophyte. Instead, the cultivate plant is an annual or biennial herb widely used as a spice and seasoning in food. A broad range of biological activities have been attributed to A. graveolens. These include antimicrobial activity, larvicidal activity, hepatoprotective activity, nematicidal and mosquito repellent potential and antihyperlipidaemic properties.
METHODS AND RESULTS:
In this study, the authors compare the composition of the volatile fractions of A. graveolens collected in natural populations in Portugal and Italy and evaluate their potential as antifungal agents.The composition of the volatile oils obtained by hydrodistillation and their antifungal activity are reported. The oils were analysed by gas chromatography-flame ionisation detector and gas chromatography-mass spectrometry methods and their composition were compared with that of the volatile extracts isolated by supercritical CO2. A chemical variability in the extracts depending on the origin of the plants and on the extraction method was observed. The results showed the presence of sedanenolide, Neocnidilide and neophytadiene as main components. The minimal inhibitory concentration (MIC) and the minimal lethal concentration were used to evaluate the antifungal activity of the oils against Candida albicans, Candida tropicalis, Candida krusei, Candida guilliermondii, Candida parapsilosis, Cryptococcus neoformans, Trichophyton rubrum, Trichophyton mentagrophytes, T. mentagrophytes var. interdigitale, Trichophyton verrucosum, Microsporum canis, Microsporum gypseum, Epidermophyton floccosum, Aspergillus niger, Aspergillus fumigatus and Aspergillus flavus. The oil from Italy rich in neophytadiene is the more active, with MIC values of 0.04-0.64 μL mL(-1).
CONCLUSIONS:
Our results show that A. graveolens volatile extracts may be useful in the clinical treatment of fungal diseases. | | Yakugaku Zasshi. 1992 Sep;112(9):638-44. | | Studies on the baths with crude drug: the effects of Senkyu extract as skin penetration enhancer.[Pubmed: 1469611] |
The effects of Senkyu (Cnidii Rhizoma and Ligustici chuanxiong Rhizoma) on the drug skin penetration were studied to clarify its effectivity as the baths.
METHODS AND RESULTS:
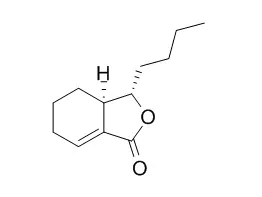
Ether and methanol extracts, and some essential oils of Senkyu (i.e. ligustilide, Neocnidilide and butylidenephthalide) enhanced remarkably the skin penetration of benzoic acid. Furthermore, an appreciable correlation between the enhancing ratio and the skin/donor partition coefficient of benzoic acid was observed.
CONCLUSIONS:
These facts suggest that the constituents of Senkyu influence the skin penetration by enhancing the partition coefficient. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)