| Structure Identification: |
| J Asian Nat Prod Res. 2012;14(5):491-5. | | A new eudesmane sesquiterpene glycosides from Liriope muscari.[Pubmed: 22423627] |
METHODS AND RESULTS:
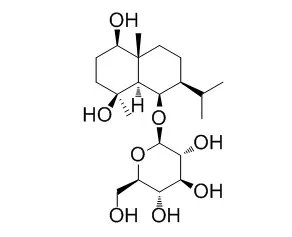
A new eudesmane sesquiterpene glycoside, ophiopogonoside B (1), along with five known compounds, Ophiopogonoside A (2), ruscogenin-1-O-[β-D-glucopyranosyl (1 → 2)]-[β-D-xylopyranosyl (1 → 3)]-β-D-fucopyranoside (3), palmitic acid (4), palmitic acid glyceride (5), and β-sitosterol-D-glucopyranoside (6),was isolated from the tuberous roots of Liriope muscari (Decn.) Bailey (Liliaceae). Their structures were confirmed by 1D and 2D NMR spectroscopy.
CONCLUSIONS:
Among them, compounds 1, 2, 4, and 5 were the first reported from the genus Liriope. Ophiopogonoside B (1) showed moderate inhibitory activity to glycogen phosphorylase a. | | J Nat Prod. 2004 Oct;67(10):1761-3. | | cis-Eudesmane sesquiterpene glycosides from Liriope muscari and Ophiopogon japonicus.[Pubmed: 15497959] |
METHODS AND RESULTS:
Two new cis-eudesmane sesquiterpene glycosides, liriopeoside A (1) and Ophiopogonoside A (2), were extracted and purified from tubers of Liriope muscari and Ophiopogon japonicus, respectively, along with three known compounds. Their structures were elucidated as 1beta,6beta-dihydroxy-cis-eudesm-3-ene-6-O-beta-D-glucopyranoside (1) and 1beta,4beta,6beta-trihydroxy-cis-eudesmane-6-O-beta-D-glucopyranoside (2) by spectral data analysis. The structure and the relative configuration of compound 1 were confirmed by X-ray crystallographic analysis.
CONCLUSIONS:
This is the first time that cis-eudesmane-type sesquiterpenes have been reported from the genera Ophiopogon and Liriope. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)