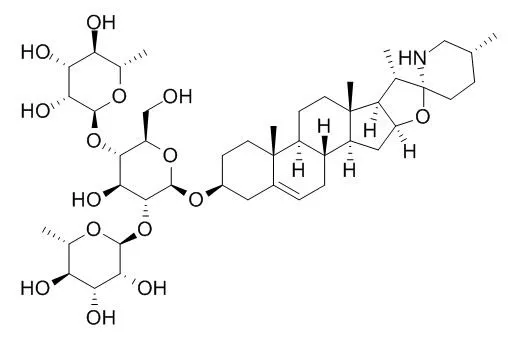
| Cell Research: |
| J Nat Med. 2014 Jan;68(1):236-41. | | Antiproliferative activity of Solanum lycocarpum alkaloidic extract and their constituents, solamargine and solasonine, in tumor cell lines.[Pubmed: 23475509] | Natural products are some of the important sources of new anticancer drugs. The Brazilian flora is considered one of the most diverse in the word, although not many large-scale pharmacological and phytochemical studies have been conducted to date.
METHODS AND RESULTS:
With this in mind, in the present study we evaluated the antiproliferative activity of Solanum lycocarpum fruit glycoalkaloid extract (SL) and its major compounds, Solamargine (SM) and solasonine (SS), against different tumor cell lines: murine melanoma (B16F10), human colon carcinoma (HT29), human breast adenocarcinoma (MCF-7), human cervical adenocarcinoma (HeLa), human hepatocellular liver carcinoma (HepG2) and human glioblastoma (MO59J, U343 and U251). The antiproliferative activity was evaluated using XTT assay and results were expressed as IC50. The most pronounced antiproliferative activity was observed for SM, with IC50 values ranging from 4.58 to 18.23 μg/mL. The lowest IC50 values were observed against HepG2, being 9.60 μg/mL for SL, 4.58 μg/mL for SM and 6.01 μg/mL for SS.
CONCLUSIONS:
Thus, SL, SM and SS demonstrated antiproliferative activity against the tumor cell lines tested, and were most effective against the HepG2 cell line. | | Life Sci. 2011 Feb 14;88(7-8):314-21. | | Solamargine induces apoptosis associated with p53 transcription-dependent and transcription-independent pathways in human osteosarcoma U2OS cells.[Pubmed: 21163271] | Solamargine, a steroidal glycoalkaloid isolated from S. incanum, has been shown to induce apoptosis in several cancer cell lines. In this study, the involvement of p53 in the pro-apoptotic action of Solamargine was investigated in human osteosarcoma U2OS cells.
METHODS AND RESULTS:
The cytotoxicity of Solamargine was evaluated by MTT assay. Solamargine-induced apoptosis was evidenced by chromatin condensation, formation of apoptotic bodies and exposure of phosphatidylserine on the extracellular surface as revealed by DAPI nuclear staining, transmission electron microscopy and flow cytometry, respectively. mRNA expressions of p53 and Bax were investigated using real time-PCR. Western blot was used to examine the changes in the expression levels of p53, Bax, Bcl-2, caspase-3, caspase-9 and cytochrome c. Subcellular localization of p53 was verified by immunofluorescence staining and cell fractionation.
Solamargine substantially reduced cell viability and induced apoptosis in osteosarcoma U2OS cells. In this connection, Solamargine increased the mRNA and protein expressions of p53 and Bax (a pro-apoptotic protein downstream to p53). The expression of Bcl-2 (an anti-apoptotic protein) was also reduced. Furthermore, Solamargine induced mitochondrial translocation of p53, loss of mitochondrial membrane potential, cytochrome c release and activation of caspase-9 and -3. p53-specific transcriptional inhibitor pifithrin-α or mitochondrial translocation inhibitor pifithrin-μ partially reversed Solamargine-induced apoptosis.
CONCLUSIONS:
Solamargine activates the mitochondria-mediated apoptotic pathway in U2OS cells via both p53 transcription-dependent and -independent mechanisms. This compound may merit further investigation as a potential therapeutic agent for the treatment of cancer. | | FEBS Lett. 2004 Nov 5;577(1-2):67-74. | | Action of solamargine on human lung cancer cells--enhancement of the susceptibility of cancer cells to TNFs.[Pubmed: 15527763 ] | Solamargine (SM), isolated from Solanum incanum herb, displayed a superior cytotoxicity in four human lung cancer cell lines.
METHODS AND RESULTS:
The half-inhibitory concentrations (IC50), of the cell viability assay for H441, H520, H661 and H69 cells were 3, 6.7, 7.2 and 5.8 microM, respectively. SM-induced apoptosis of these cells by PS externalization in a dose-dependent manner and increased sub-G1 fraction were observed. Quenching of the expression of tumor necrosis factor receptors (TNFRs) during the progress of human lung carcinogenesis has been previously reported. SM may induce cell apoptosis via modulating the expression of TNFRs and their subsequent TRADD/FADD signal cascades. Subsequently, SM treatment increased the binding activities of TNF-alpha and TNF-beta to the lung cancers, and the intrinsic TNFs-resistant cancer cells became susceptible to TNF-alpha and -beta. In addition, SM caused release of cytochrome c, downregulation of anti-apoptotic Bcl-2 and Bcl-xL, increase of caspase-3 activity, and DNA fragmentation.
CONCLUSIONS:
Thus, SM could modulate the expressions of TNFRs and Bcl-2, and might be a potential anticancer agent for TNFs and Bcl-2 related resistance of human lung cancer cells. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)