| J Agric Food Chem. 2012 Sep 12;60(36):9292-7. |
| Competition between ascorbate and glutathione for the oxidized form of methylated quercetin metabolites and analogues: tamarixetin, 4'O-methylquercetin, has the lowest thiol reactivity.[Pubmed: 22860763] |
Quercetin (Q) is a bioactive compound with excellent antioxidant activity. However, the thiol reactivity of its oxidation product (oxQ) forms a disadvantage. The aim of the present study was to decrease this thiol toxicity.
METHODS AND RESULTS:
We found that methylated Q metabolites displayed lower thiol reactivity than Q. The most effective was Tamarixetin, 4'O-methylquercetin (4'MQ), that has a corresponding oxidation product (ox4'MQ) with thiol reactivity 350 times lower than oxQ. The endogenous metabolism of Q to 4'MQ might be a physiological way to safely benefit from the antioxidant potential of Q in vivo.
CONCLUSIONS:
Our results were explained with Pearson's HSAB concept and corroborated by quantum molecular calculations that revealed a strong correlation between the relative thiol reactivity and the lowest unoccupied molecular orbital (LUMO). The polarity of the molecule and the π-π interaction between the AC- and the B-ring appeared to determine the LUMO and the thiol reactivity of the oxidation product. |
| Brit. J. Pharmacol., 2000, 131: U11-U11. |
| Vasodilator effects of quercetin and its metabolites, isorhamnetin and tamarixetin, in rat isolated vessels.[Reference: WebLink] |
METHODS AND RESULTS:
Vasodilator effects of quercetin and its metabolites, isorhamnetin and Tamarixetin, in rat isolated vessels. |
| J Mol Histol . 2019 Aug;50(4):343-354. |
| Tamarixetin protects against cardiac hypertrophy via inhibiting NFAT and AKT pathway[Pubmed: 31111288] |
| Abstract
Cardiac hypertrophy is a compensatory response in reaction to mechanical load that reduces wall stress by increasing wall thickness. Chronic hypertrophic remodeling involves cardiac dysfunction that will lead to heart failure and ultimately death. Studies have been carried out on cardiac hypertrophy for years, whereas the mechanisms have not been well defined. Tamarixetin (TAM), a natural flavonoid derivative of quercetin, have been demonstrated possessing anti-oxidative and anti-inflammatory effects on multiple diseases. However, little is known about the function of TAM on the development of cardiac hypertrophy. Here, we found TAM could alleviate pressure-overload-induced cardiac hypertrophy in transverse aortic constriction (TAC) mouse model, assessed by ventricular weight/body weight, lung weight/body weight, echocardiographic parameters, as well as myocyte cross-sectional area and the expression of ANP, BNP and Myh7. In vitro, TAM showed a dose dependent inhibitory effect on phenylephrine-induced hypertrophy in H9c2 cardiomyocytes. Furthermore, TAM reversed cardiac remodeling of stress overloaded heart by suppressing apoptosis and the expression of fibrotic-related genes, reduced oxidative stress and ROS production both in vivo and in vitro. In addition, TAM could negatively modulate TAC-induced nuclear translocation of NFAT and the activation of PI3K/AKT signaling pathways. Therefore, these data indicate for the first time that TAM has a protective effect on experimental cardiac hypertrophy and might be a novel candidate for the treatment of cardiac hypertrophy in clinic.
Keywords: Cardiac hypertrophy; Oxidative stress; Remodeling; Tamarixetin. |
| J Nat Prod . 2018 Jun 22;81(6):1435-1443. |
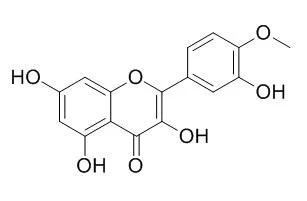
| CFN97027[Pubmed: 29851490] |
| Abstract
Sepsis is a systemic inflammatory response to pathogenic infection that currently has no specific pharmaceutical interventions. Instead, antibiotics administration is considered the best available option, despite increasing drug resistance. Alternative strategies are therefore urgently required to prevent sepsis and strengthen the host immune system. One such option is Tamarixetin (4'- O-methylquercetin), a naturally occurring flavonoid derivative of quercetin that protects against inflammation. The purpose of this study was to determine whether the anti-inflammatory effects of Tamarixetin protect against the specific inflammatory conditions induced in lipopolysaccharide (LPS) or Escherichia coli K1 models of sepsis. Our study showed that Tamarixetin reduced the secretion of various inflammatory cytokines by dendritic cells after activation with LPS. It also promoted the secretion of the anti-inflammatory cytokine interleukin (IL)-10 and specifically increased the population of IL-10-secreting immune cells in LPS-activated splenocytes. Tamarixetin showed general anti-inflammatory effects in mouse models of bacterial sepsis and decreased bacteria abundance and endotoxin levels. We therefore conclude that Tamarixetin has superior anti-inflammatory properties than quercetin during bacterial sepsis. This effect is associated with an increased population of IL-10-secreting immune cells and suggests that Tamarixetin could serve as a specific pharmaceutical option to prevent bacterial sepsis. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)