| Description: |
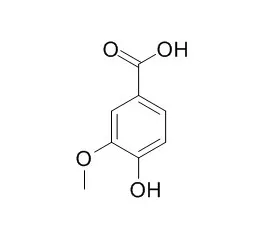
Vanillic acid is a flavoring agent which has hepatoprotective, free radical scavenging, antioxidant and anti-inflammatory properties. It exerts protective effects in isoproterenol induced cardiotoxic rats, has inhibitory effect on methylglyoxal-mediated glycation in apoptotic Neuro-2A cells via inhibition of glycation mechanisms including ROS, p38 and JNK, PKC and p47(phox).
|
| Targets: |
TNF-α | ROS | p38MAPK | JNK | PKC | p47(phox) | IL Receptor | Fas | Caspase |
| In vitro: |
| Neurotoxicology. 2008 Nov;29(6):1016-22. | | Inhibitory effect of vanillic acid on methylglyoxal-mediated glycation in apoptotic Neuro-2A cells[Pubmed: 18706441] | Methylglyoxal is a reactive dicarbonyl compound generated as an intermediate of glycolysis during the physical glycation in the diabetic condition. It is considered to be a potent precursor of advanced glycation end products (AGEs) formation. Methylglyoxal itself and methylglyoxal-derived AGEs have been commonly implicated in the development of diabetic neuropathy. Our previous study indicated that Vanillic acid showed an inhibitory effect against methylglyoxal-mediated Neuro-2A cell apoptosis, suggesting that Vanillic acid might possess cytoprotective properties in the prevention of diabetic neuropathy complication.
METHODS AND RESULTS:
In this study, the effects of Vanillic acid on the methylglyoxal-mediated glycation system involved in the progression of Neuro-2A cell apoptosis were further investigated. Our findings indicated that methylglyoxal-induced Neuro-2A cell apoptosis was mediated through the possible glycation mechanism of oxidative stress, activation of the MAPK signaling pathway (p38 and JNK) and oxidation-sensitive protein expression (PKC and p47(phox)) and methylglyoxal-derived N-epsilon-(carboxymethyl)lysine (CML) formation. Vanillic acid, however, suppressed methylglyoxal-induced Neuro-2A cell apoptosis via inhibition of glycation mechanisms including ROS, p38 and JNK, PKC and p47(phox), and methylglyoxal-derived CML formation.
CONCLUSIONS:
In the present study, we established the first evidence that Vanillic acid might contribute to the prevention of the development of diabetic neuropathy by blocking the methylglyoxal-mediated intracellular glycation system. | | Sci Rep . 2017 Jan 18;7:40753. | | Vanillic acid attenuates Aβ 1-42-induced oxidative stress and cognitive impairment in mice[Pubmed: 28098243] | | Abstract
Increasing evidence demonstrates that β-amyloid (Aβ) elicits oxidative stress, which contributes to the pathogenesis and disease progression of Alzheimer's disease (AD). The aims of the present study were to determine and explore the antioxidant nature and potential mechanism of Vanillic acid (VA) in Aβ1-42-induced oxidative stress and neuroinflammation mediated cognitive impairment in mice. An intracerebroventricular (i.c.v.) injection of Aβ1-42 into the mouse brain triggered increased reactive oxygen species (ROS) levels, neuroinflammation, synaptic deficits, memory impairment, and neurodegeneration. In contrast, the i.p. (intraperitoneal) administration of VA (30 mg/kg, for 3 weeks) after Aβ1-42-injection enhanced glutathione levels (GSH) and abrogated ROS generation accompanied by an induction of the endogenous nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase 1 (HO-1) via the activation of Akt and glycogen synthase kinase 3β (GSK-3β) in the brain mice. Additionally, VA treatment decreased Aβ1-42-induced neuronal apoptosis and neuroinflammation and improved synaptic and cognitive deficits. Moreover, VA was nontoxic to HT22 cells and increased cell viability after Aβ1-42 exposure. To our knowledge, this study is the first to reveal the neuroprotective effect of VA against Aβ1-42-induced neurotoxicity. Our findings demonstrate that VA could potentially serve as a novel, promising, and accessible neuroprotective agent against progressive neurodegenerative diseases such as AD. |
|
| In vivo: |
| Eur J Pharmacol. 2011 Oct 1;668(1-2):233-40. | | Protective effects of vanillic acid on electrocardiogram, lipid peroxidation, antioxidants, proinflammatory markers and histopathology in isoproterenol induced cardiotoxic rats.[Pubmed: 21763302 ] | Myocardial infarction affects a large proportion in the world. This study aims to evaluate the protective effects of Vanillic acid in isoproterenol induced cardiotoxic rats.
METHODS AND RESULTS:
Male Wistar rats were pretreated with Vanillic acid (5mg and 10mg/kg) daily for 10 days. After pretreatment, isoproterenol (100mg/kg) was subcutaneously injected to rats at an interval of 24h for 2 days to induce cardiotoxicity. Isoproterenol induced cardiotoxic rats showed significant increase in serum cardiac troponins, heart lipid peroxidation and significant decrease in the heart antioxidants. In addition, isoproterenol induced group electrocardiogram showed an elevation in ST segments and increased expressions of interleukin-1β, interleukin-6 and tumor necrosis factor-α genes in the myocardium. Pretreatment with Vanillic acid showed significant protective effects on cardiac troponins, lipid peroxidation, antioxidant system, electrocardiogram and expressions of interleukin-1β, interleukin-6 and tumor necrosis factor-α gene in the heart of isoproterenol induced cardiotoxic rats. Histopathology of myocardium correlated with these biochemical findings. The in vitro study also revealed that Vanillic acid is a potent free radical scavenger. Thus, Vanillic acid exerts protective effects in isoproterenol induced cardiotoxic rats due to its free radical scavenging, antioxidant and anti-inflammatory properties.
CONCLUSIONS:
Our study also showed that pretreatment with Vanillic acid at the dose of 10mg/kg was highly effective than 5mg/kg. | | Biol Pharm Bull. 2010;33(6):983-7. | | Hepatoprotective effect of syringic acid and vanillic acid on CCl4-induced liver injury.[Pubmed: 20522963] | The mycelia of the edible mushroom Lentinula edodes can be cultured in solid medium containing lignin, and the hot-water extracts (L.E.M.) is commercially available as a nutritional supplement. During the cultivation, phenolic compounds, such as syringic acid and Vanillic acid, were produced by lignin-degrading peroxidase secreted from L. edodes mycelia. Since these compounds have radical scavenging activity, we examined their protective effect on oxidative stress in mice with CCl(4)-induced liver injury.
METHODS AND RESULTS:
We examined the hepatoprotective effect of syringic acid and Vanillic acid on CCl(4)-induced chronic liver injury in mice. The injection of CCl(4) into the peritoneal cavity caused an increase in the serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels. The intravenous administration of syringic acid and Vanillic acid significantly decreased the levels of the transaminases. Four weeks of CCl(4) treatment caused a sufficiently excessive deposition of collagen fibrils. An examination of Azan-stained liver sections revealed that syringic acid and Vanillic acid obviously suppressed collagen accumulation and significantly decreased the hepatic hydroxyproline content, which is the quantitative marker of fibrosis. Both of these compounds inhibited the activation of cultured hepatic stellate cells, which play a central role in liver fibrogenesis, and maintained hepatocyte viability.
CONCLUSIONS:
These data suggest that the administration of syringic acid and Vanillic acid could suppress hepatic fibrosis in chronic liver injury. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)