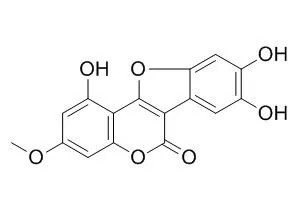
| Description: |
Wedelolactone is a potent Î2-arrestin-biased G protein-coupled receptor-35 (GPR35) agonist, GPR35 has been shown to be a target of the asthma drugs cromolyn disodium and nedocromil sodium. Wedelolactone has anti-inflammatory, growth inhibitory, anti-cancer, anti-fibrotic, and pro-apoptotic effects. Wedelolactone stimulates ER genomic and non-genomic signalling pathways; it can significantly inhibit the activation of LX-2 cells, the underlying mechanisms of which included inducing Bcl-2 family involved apoptosis, up-regulating phosphorylated status of ERK and JNK expressions, and inhibiting NF-κB mediated activity.
|
| In vitro: |
| J Ethnopharmacol. 2014 Nov 18;157:206-11. | | Inhibitory effect of Ecliptae herba extract and its component wedelolactone on pre-osteoclastic proliferation and differentiation.[Pubmed: 25267578] | Ecliptae herba, also known as "Mo-Han-Lian", has long been used in China to nourish Kidney and thereafter strengthen bones. Accumulating evidence indicates that extracts of Ecliptae herba have antiosteoporotic effect. However, the effective compounds and cellular mode of action are still unclear. To investigate the effect of ethyl acetate extract of Ecliptae herba (EAE) and its component Wedelolactone on proliferation and differentiation of preosteoclastic RAW264.7 cells as well as proliferation of bone marrow stromal cells (BMSC).
METHODS AND RESULTS:
RAW264.7 and BMSC were examined for proliferation by a 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) method. Tartrate-resistant acid phosphatase (TRAP) activity of RAW264.7 was measured by using p-nitrophenyl sodium phosphate (pNPP) assay after the cells were treated with 30ng/ml receptor activator for nuclear factor-κ B ligand (RANKL) plus various concentrations of EAE, Wedelolactone or alendronate. The formation of multinucleated TRAP-positive RAW264.7 cells was observed by using a TRAP-staining kit.
Treatment of RAW264.7 cells with EAE at high doses (20μg/ml and 40μg/ml) or Wedelolactone at 10μg/ml resulted in a decrease in proliferation of RAW264.7 cells. Low doses of EAE (5, 10μg/ml) and Wedelolactone (2.5μg/ml) inhibited RANKL-induced TRAP activity by 20.3%, 37.9%, and 48.3%. The inhibitory effect of Wedelolactone is more potent than that of alendronate, an anti-resorptive drug. Morphological changes revealed that 5μg/ml EAE and 2.5μg/ml Wedelolactone reduced the number of multinucleated osteoclast-like cells. At the high doses, EAE (20μg/ml) and Wedelolactone (10μg/ml) inhibited the growth of BMSC.
CONCLUSIONS:
EAE and its component Wedelolactone inhibited osteoclast RAW264.7 proliferation and differentiation at the low doses, but at the high doses, showed cytotoxic effect on BMSC. These results indicated that EAE and wedelolatone might be potential alternative therapy for osteoporosis. | | J Cell Biochem. 2012 Nov;113(11):3436-45. | | Wedelolactone inhibits adipogenesis through the ERK pathway in human adipose tissue-derived mesenchymal stem cells.[Pubmed: 22678810] | Wedelolactone is an herbal medicine that is used to treat septic shock, hepatitis and venom poisoning. Although in differentiated and cancer cells, Wedelolactone has been identified as anti-inflammatory, growth inhibitory, and pro-apoptotic, the effects of Wedelolactone on stem cell differentiation remain largely unknown.
METHODS AND RESULTS:
Here, we report that Wedelolactone inhibits the adipogenic differentiation of human adipose tissue-derived mesenchymal stem cells (hAMSCs). Wedelolactone reduced the formation of lipid droplets and the expression of adipogenesis-related proteins, such as CCAAT enhancer-binding protein-α (C/EBP-α), peroxisome proliferator-activated receptor-γ (PPAR-γ), lipoprotein lipase (LPL), and adipocyte fatty acid-binding protein aP2 (aP2). Wedelolactone mediated this process by sustaining ERK activity. In addition, inhibition of ERK activity with PD98059 resulted in reversion of the Wedelolactone-mediated inhibition of adipogenic differentiation.
CONCLUSIONS:
Taken together, these results indicate that Wedelolactone inhibits adipogenic differentiation through ERK pathway and suggest a novel inhibitory effect of Wedelolactone on adipogenic differentiation in hAMSCs. | | Oncotarget. 2015 May 30;6(15):13049-59. | | Wedelolactone disrupts the interaction of EZH2-EED complex and inhibits PRC2-dependent cancer.[Pubmed: 25944687] | Polycomb repressive complex 2 (PRC2), which is responsible for the trimethylation of H3K27 (H3K27me3), plays a part in tumorigenesis, development and/or maintenance of adult tissue specificity. The pivotal role of PRC2 in cancer makes it a therapeutic target for epigenetic cancer therapy. However, natural compounds targeting the enhancer of zeste homolog 2 (EZH2) - embryonic ectoderm development (EED) interaction to disable PRC2 complex are scarcely reported.
METHODS AND RESULTS:
Here, we reported the screening and identification of natural compounds which could disrupt the EZH2-EED interaction. One of these compounds, Wedelolactone, binds to EED with a high affinity (KD = 2.82 μM), blocks the EZH2-EED interaction in vitro, induces the degradation of PRC2 core components and modulates the expression of detected PRC2 downstream targets and cancer-related genes. Furthermore, some PRC2-dependent cancer cells undergone growth arrest upon treatment with Wedelolactone.
CONCLUSIONS:
Thus, Wedelolactone and its derivatives which target the EZH2-EED interaction could be candidates for the treatment of PRC2-dependent cancer. |
|
| In vivo: |
| Sci Rep . 2016 Aug 25;6:32260. | | Wedelolactone enhances osteoblastogenesis by regulating Wnt/β-catenin signaling pathway but suppresses osteoclastogenesis by NF-κB/c-fos/NFATc1 pathway[Pubmed: 27558652] | | Abstract
Bone homeostasis is maintained by formation and destruction of bone, which are two processes tightly coupled and controlled. Targeting both stimulation on bone formation and suppression on bone resorption becomes a promising strategy for treating osteoporosis. In this study, we examined the effect of Wedelolactone, a natural product from Ecliptae herba, on osteoblastogenesis as well as osteoclastogenesis. In mouse bone marrow mesenchymal stem cells (BMSC), Wedelolactone stimulated osteoblast differentiation and bone mineralization. At the molecular level, Wedelolactone directly inhibited GSK3β activity and enhanced the phosphorylation of GSK3β, thereafter stimulated the nuclear translocation of β-catenin and runx2. The expression of osteoblastogenesis-related marker gene including osteorix, osteocalcin and runx2 increased. At the same concentration range, Wedelolactone inhibited RANKL-induced preosteoclastic RAW264.7 actin-ring formation and bone resorption pits. Further, Wedelolactone blocked NF-kB/p65 phosphorylation and abrogated the NFATc1 nuclear translocation. As a result, osteoclastogenesis-related marker gene expression decreased, including c-src, c-fos, and cathepsin K. In ovariectomized mice, administration of Wedelolactone prevented ovariectomy-induced bone loss by enhancing osteoblast activity and inhibiting osteoclast activity. Together, these data demonstrated that Wedelolactone facilitated osteoblastogenesis through Wnt/GSK3β/β-catenin signaling pathway and suppressed RANKL-induced osteoclastogenesis through NF-κB/c-fos/NFATc1 pathway. These results suggested that wedelolacone could be a novel dual functional therapeutic agent for osteoporosis. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)