- ChemFaces is a professional high-purity natural products manufacturer.
- Product Intended Use
- 1. Reference standards
- 2. Pharmacological research
- 3. Inhibitors
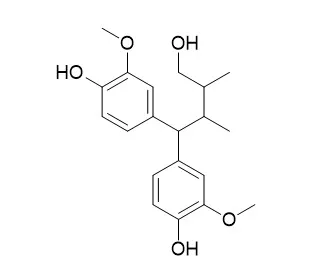
4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol
ChemFaces products have been cited in many studies from excellent and top scientific journals
Contact Us
Order & Inquiry & Tech Support
Tel: (0086)-27-84237683
Tech: service@chemfaces.com
Order: manager@chemfaces.com
Address: 176, CheCheng Eest Rd., WETDZ, Wuhan, Hubei 430056, PRC
How to Order
Orders via your E-mail:
1. Product number / Name / CAS No.
2. Delivery address
3. Ordering/billing address
4. Contact information
Order: manager@chemfaces.com
Delivery time
Delivery & Payment method
1. Usually delivery time: Next day delivery by 9:00 a.m. Order now
2. We accept: Wire transfer & Credit card & Paypal
Citing Use of our Products
* Packaging according to customer requirements(5mg, 10mg, 20mg and more). We shipped via FedEx, DHL, UPS, EMS and others courier.
According to end customer requirements, ChemFaces provide solvent format. This solvent format of product intended use: Signaling Inhibitors, Biological activities or Pharmacological activities.
| Size /Price /Stock |
10 mM * 1 mL in DMSO / $312.8 / In-stock |
Other Packaging |
*Packaging according to customer requirements(100uL/well, 200uL/well and more), and Container use Storage Tube With Screw Cap |
More articles cited ChemFaces products.
- Biochem Pharmacol.2017, 130:10-20
- Kor. J. Pharmacogn.2016, 47(1):62-72
- J of Physics Conference Series...2019...
- Front Plant Sci.2020, 11:630.
- J of l. Chroma.&Related Tech...2020...
- J Sep Sci.2019, 42(21):3352-3362
- BMC Plant Biol.2018, 18(1):122
- Molecules.2020, 25(11):2599.
- BMC Pharmacol Toxicol.2018, 19(1):5
- Molecules.2015, 20(11):20014-30
- Chem Biol Interact.2019, 298:1-7
- Institute of Food Science & Techn...2021...
- Food Chem.2017, 221:1135-1144
- J Ethnopharmacol.2017, 206:73-77
- J Agric Food Chem....2021...
- Molecules.2016, 21(10)
- Molecules.2021, 26(19):6032.
- The Journal of Animal & Plant Sci...2020...
- Pharmaceutics.2021, 13(11):1839.
- Korean J. Food Preserv. ...2021...
- Int J Mol Sci.2015, 16(1):1232-51
- Processes2021, 9(1), 153;
- J Ethnopharmacol.2022, 291:115159.
- More...
Our products had been exported to the following research institutions and universities, And still growing.
- University of Sao Paulo (Brazil)
- Kazusa DNA Research Institute (Japan)
- Cornell University (USA)
- Heinrich-Heine-University Düss... (Germany)
- University of Maryland School o... (USA)
- Universidad de Buenos Aires (Argentina)
- Ateneo de Manila University (Philippines)
- University of Canterbury (New Zealand)
- Universidad de Antioquia (Colombia)
- University of Melbourne (Australia)
- Nanjing University of Chinese M... (China)
- More...
- Aquaculture2019, 510:392-399
- Korean J of Pharmacognosy2020, 51,49-54.
- Antioxidants (Basel).2021, 10(9):1487.
- Earth Environ. Sci. 2021, 905:012080.
- Biochem Biophys Res Commun.2018, 495(1):1271-1277
- Appl. Sci.2021, 11(19),9343.
- Drug Test Anal.2018, 10(10):1579-1589
- Cancers (Basel).2023, 15(1):37.
- Molecules.2021, 26(9):2526.
- Nutrients.2017, 10(1)
Related Screening Libraries
| Size /Price /Stock |
10 mM * 100 uL in DMSO / Inquiry / In-stock
10 mM * 1 mL in DMSO / Inquiry / In-stock
|
Related Libraries |
|
| Description: |
4,4-di(4-Hydroxy-3-methoxyphenly)-2,3-dimethylbutanol has antioxidant and potential cytotoxic abilities, it also shows inhibition against HIV-1 integrase. |
| Targets: |
HIV |
| In vitro: |
| Biol Res. 2009;42(3):351-6. | | Antioxidant isolated from Schisandra propinqua (Wall.) Baill.[Pubmed: 19915743 ] | Schisandra propinqua (Wall.) Baill.(Schisandraceae) is widely used as a Chinese folk medicine.
METHODS AND RESULTS:
In this study, activity-guided fractionation of the ethanol extract from the stem of Schisandra propinqua led to the isolation of four extracts. Subsequently, a neolignan
4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol was isolated from the EtOAc part of the stem of Schisandra propinqua, the free radical scavenging activities of which were researched in vitro. The present work demonstrated that extracts and pure compound possessed scavenging activities to DPPH, superoxide anions and hydroxy radical, and could depress lipid peroxidation reaction induced by oxygen radical produced by the Fe2+/cysteine system in vitro.
CONCLUSIONS:
This suggests that the traditional application of Schisandra propinqua in China may be related to its antioxidant activities, and the EtOAc part of the stems of Schisandra propinqua can be utilized as an effective source of antioxidants. |
|
4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol Description
| Source: |
The herbs of Schisandra propinqua |
| Solvent: |
Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| Storage: |
Providing storage is as stated on the product vial and the vial is kept tightly sealed, the product can be stored for up to 24 months(2-8C).
Wherever possible, you should prepare and use solutions on the same day. However, if you need to make up stock solutions in advance, we recommend that you store the solution as aliquots in tightly sealed vials at -20C. Generally, these will be useable for up to two weeks. Before use, and prior to opening the vial we recommend that you allow your product to equilibrate to room temperature for at least 1 hour.
Need more advice on solubility, usage and handling? Please email to: service@chemfaces.com
|
| After receiving: |
The packaging of the product may have turned upside down during transportation, resulting in the natural compounds adhering to the neck or cap of the vial. take the vial out of its packaging and gently shake to let the compounds fall to the bottom of the vial. for liquid products, centrifuge at 200-500 RPM to gather the liquid at the bottom of the vial. try to avoid loss or contamination during handling. |
ChemFaces New Products and Compounds
Recently, ChemFaces products have been cited in many studies from excellent and top scientific journals

Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.
IF=36.216(2019)PMID: 29328914
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.
IF=22.415(2019)PMID: 32004475
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.
IF=14.548(2019)PMID: 29149595
ACS Nano. 2018 Apr 24;12(4): 3385-3396. doi: 10.1021/acsnano.7b08969.
IF=13.903(2019)PMID: 29553709
Nature Plants. 2016 Dec 22;3: 16206. doi: 10.1038/nplants.2016.205.
IF=13.297(2019)PMID: 28005066
Sci Adv. 2018 Oct 24;4(10): eaat6994. doi: 10.1126/sciadv.aat6994.
IF=12.804(2019)PMID: 30417089
Calculate Dilution Ratios(Only for Reference)
|
1 mg |
5 mg |
10 mg |
20 mg |
25 mg |
| 1 mM |
2.8868 mL |
14.4342 mL |
28.8684 mL |
57.7367 mL |
72.1709 mL |
| 5 mM |
0.5774 mL |
2.8868 mL |
5.7737 mL |
11.5473 mL |
14.4342 mL |
| 10 mM |
0.2887 mL |
1.4434 mL |
2.8868 mL |
5.7737 mL |
7.2171 mL |
| 50 mM |
0.0577 mL |
0.2887 mL |
0.5774 mL |
1.1547 mL |
1.4434 mL |
| 100 mM |
0.0289 mL |
0.1443 mL |
0.2887 mL |
0.5774 mL |
0.7217 mL |
* Note: If you are in the process of experiment, it's need to make the dilution ratios of the samples. The dilution data of the sheet for your reference. Normally, it's can get a better solubility within lower of Concentrations.
| Kinase Assay: |
| Yao Xue Xue Bao. 2010 Feb;45(2):235-40. | | Effective components against HIV-1 replicative enzymes isolated from plants.[Pubmed: 21351433] | Plant active components characterized of many different structures and activities on multiple targets, have made them to be the important sources of inhibitors on HIV-1.
METHODS AND RESULTS:
For finding leading compounds with new structure against HIV-1, three key HIV-1 replicative enzymes (reverse transcriptase, protease and integrase) were used as screening models. The in vitro activities of 45 plant derived components isolated from Schisandraceae, Rutaceae and Ranunculaceae were reported. Within twelve triterpene components isolated, eight compounds were found to inhibit HIV-1 protease, in these eight active compounds, kadsuranic acid A (7) and nigranoic acid (8), inhibited both HIV-1 protease and integrase; Among fifteen lignans, meso-dihydroguaiaretic acid (15) and kadsurarin (16) were active on HIV-1 reverse transcriptase, and
4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol(13) active on HIV-1 integrase.
CONCLUSIONS:
All of the six alkaloids, seven flavones, and five others compounds were not active or only with low activities against HIV-1 replicative enzymes. Further studies of the triterpene components showing strong inhibitory activities on HIV-1 were warranted. |
|
| Structure Identification: |
| Journal of Integrative Plant Biology, 2006, 48(12):1493-1497. | | A Cytotoxic Neolignan from Schisandra propinqua (Wall.) Baill.[Reference: WebLink] | In the course of our study of bioactive natural products from Schisandra plants, we isolated a neolignan from an EtOAc extract of the stems of Schisandra propinqua (Wall.) Baill.
METHODS AND RESULTS:
The structure of the new compound was determined to be
4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol(compound 1) on the basis of 1H- and 13C-NMR spectra and 2D NMR methods. Eight known compounds, compounds 2-9, were also isolated and identified, of which compounds 3, 4, 6 and 9 were isolated for the first time from this plant. In addition, compounds 1-4 were evaluated for cytotoxicity by an 3-(4,5-dimethyl-2 thiazoyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay.
CONCLUSIONS:
Compound 1 showed significant potential cytotoxic ability in the bioassay. |
|
Tags: buy 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol | 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol supplier | purchase 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol | 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol cost | 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol manufacturer | order 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol | 4,4-di(4-hydroxy-3-methoxyphenly)-2,3-dimethylbutanol distributor






 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)