| In vitro: |
| Food Chem. 2014 Dec 1;164:150-7. | | Structural characterisation and antioxidant activity evaluation of phenolic compounds from cold-pressed Perilla frutescens var. arguta seed flour.[Pubmed: 24996318] |
METHODS AND RESULTS:
A total of 11 phenolic compounds, as well as sucrose (12) and tryptophan (13), were isolated from cold-pressed Perilla frutescens var. arguta seed flour using column chromatography, and their chemical structures were identified as 3'-dehydroxyl-rosmarinic acid-3-o-glucoside (1), rosmarinic acid-3-o-glucoside (2), rosmarinic acid (3), rosmarinic acid methyl ester (4), luteolin (5), luteolin-5-o-glucoside (6), apigenin (7), caffeic acid (8), caffeic acid-3-o-glucoside (9), vanillic acid (10) and Cimidahurinine (11) using NMR and time-of-flight mass spectrometry.
CONCLUSIONS:
Of these components, compound 1 is novel, and this is the first report of compounds 10 and 11 in perilla seeds.
HPLC quantification combined with antioxidant activity evaluation revealed that rosmarinic acid and rosmarinic acid-3-o-glucoside were the dominant phenolic antioxidants with strong antioxidant activities. | | J Asian Nat Prod Res. 2005 Oct;7(5):695-9. | | Triterpenoid glycoside from Cimicifuga racemosa.[Pubmed: 16176901] |
METHODS AND RESULTS:
One new triterpene glycoside, cimiracemoside (I), and 14 known triterpene glycosides have been isolated from the rhizome extracts of black cohosh (Cimicifuga racemosa).
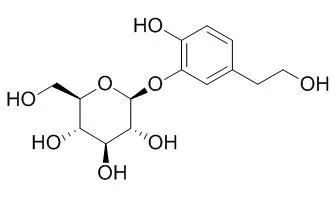
On the basis of spectral and chemical evidence, the structure of the new compound was elucidated to be 12β-acetoxycimigenol-3-O-β-d-xylopranoside (1), and the known compounds were identified to be 25—acetylcimigenol xyloside (2), cimigenol-3-O-β-d-xylopyranoside (3), acetin (4), 27-deoxyacetin (5), cimicifugoside H-1 (6), 23-O-acetylshengmanol 3-O-β-d-xylopranoside (7), foetidinol-3-O-β-xyloside (8), cimicifugoside H-2 (9), 25-O-methylcimigenol xyloside (10), 21-hydroxycimigenol-3-O-β-d-xylopyranoside (11), 24-epi-7,8—didehydrocimigenol-3-xyloside (12), Cimidahurinine (13), cimidahurine (14) and cimifugin (15).
CONCLUSIONS:
The compounds 1–5, 14, and 15 showed weak antibacterial activities in the agar diffusion assay. | | Rsc Adv., 2015, 5(129):106431-8. | | A dual-fluorescent whole-well imaging approach for screening active compounds against doxorubicin-induced cardiotoxicity from natural products[Reference: WebLink] | Doxorubicin (DOX) is an effective chemotherapy drug for various types of cancer. However, acute and chronic cardiotoxicity of DOX hamper its clinical application. There is a great demand for the discovery of drugs against DOX-induced cardiotoxicity. This paper proposed a dual fluorescence cellular imaging assay for screening active compounds against DOX toxicity.
METHODS AND RESULTS:
Whole-well fluorescence images of cells in 96-well microplates were automatically acquired and reconstructed by using a fluorescence microscope coupled with a computer-controlled moving stage. DOX-injured cardiomyocytes were labeled with two fluorescent probes, namely, fluorescein diacetate and Hoechst 33342, to determine cell viability and apoptosis. The linear range and sensitivity of the proposed approach were evaluated and validated by a known active compound, rutin. The proposed approach was also successfully applied in screening active compounds from a clinically used herbal medicine ZhenQiFuZheng granule (ZQFZ), which consisted of two herbs, Astragalus membranaceus (Fisch.) Bunge and Ligustrum lucidum. Five active components were found and were further analyzed by liquid chromatography coupled with mass spectrometry. Hydroxytyrosol, neonuezhenide, salidroside, and Cimidahurinine attenuated DOX-induced cardiotoxicity in a dose-dependent manner with EC50 values of 198.0, 260.4, 621.7, and 45.79 μM, respectively.
CONCLUSIONS:
Western blot results indicated that these active compounds protected against cardiotoxicity by decreasing reactive oxygen species (ROS) accumulation and downregulating apoptosis-related Bax/Bcl-2 proteins. The proposed approach is efficient in screening active compounds from natural products and other complex mixtures. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)