| In vitro: |
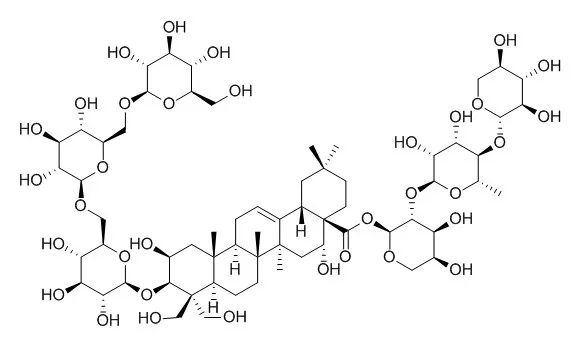
| Natural Product Sciences, 2008, 14(4):274-280. | | Compositional analysis of major saponins and anti-inflammatory activitiy of steam-processed platycodi radix under pressure.[Reference: WebLink] | Platycosides are the saponins in Platycodi Radix and they have several beneficial effects such as anti-inflammatory and anti-obesity activities. This study was designed to determine the changes in the saponin composition in Platycodi Radix (platycosides) after being processed under steam and pressure and to investigate the anti-inflammatory effects of their extracts.
METHODS AND RESULTS:
The change of the platycoside compositions was investigated after 1, 2, 3, 6 and 9h heat processing of Platycodi Radices by using HPLC coupled with an evaporative light scattering detection (ELSD) system. After heat treatment (125 °C, 1, 2, 3, 6 and 9 h), the contents of several platycosides such as platycoside E, platycodin D3, platycodin D, polygalacin D, and platycodin A decreased as the processing time was longer. While the total contents of the saponins decreased, the contents of deapi-forms of Deapi-platycoside E, deapi-platycodin D3, and deapi-platycodin D increased relatively. These results indicate that the linkage between apiose and xylose located at C-28 is labile to heat and pressure. The LPS-induced iNOS inhibitory activities of the samples treated for 1 and 2 hours were enhanced and after then, the activities were reduced.
CONCLUSIONS:
These results suggested that heat treatment of the samples affect the content of the total saponins and the saponin content may be the important criteria representing the anti-inflammatory activity. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)