| In vitro: |
| Zhongguo Zhong yao za zhi,2013, 38(14):2321-2324. | | Chemical constituents from stems of Brucea mollis and their cytotoxic activity.[Reference: WebLink] |
METHODS AND RESULTS:
Ten compounds were isolated from the stems of Brucea mollis by various chromatographic techniques such as column chromatography on silica gel and Sephadex LH-20, and preparative HPLC, and their structures were elucidated as deacetylated isobrucein B (1), indaquassin X (2), cleomiscosin A (3), cleomiscosin B (4), (+)-lyoniresinol (5), (+)-Epipinoresinol(6), (+)-pinoresinol (7), (+)-syringaresinol (8), 4,5-dihydroblumenol A (9) and adenosine (10) on the basis of spectroscopic data analysiS. All compounds were obtained from this plant for the first time, moreover, compound 1 was a new natural product.
CONCLUSIONS:
Compound 2 showed significant cytotoxic activities against the human cell lines HT-29, HepG2, BGC-823 and SKOV3 with IC50 values of 0.84-3.97 micromol x L(-1). | | Plant Cell Physiol . 2018 Nov 1;59(11):2278-2287. | | Formation of a Methylenedioxy Bridge in (+)-Epipinoresinol by CYP81Q3 Corroborates with Diastereomeric Specialization in Sesame Lignans[Pubmed: 30085233] | | Abstract
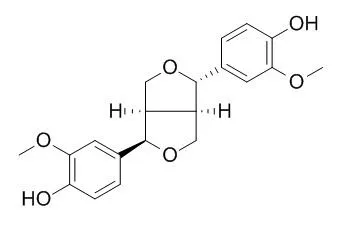
Plant specialized metabolites are often found as lineage-specific diastereomeric isomers. For example, Sesamum alatum accumulates the specialized metabolite (+)-2-episesalatin, a furofuran-type lignan with a characteristic diastereomeric configuration rarely found in other Sesamum spp. However, little is known regarding how diastereomeric specificity in lignan biosynthesis is implemented in planta. Here, we show that S. alatum CYP81Q3, a P450 orthologous to S. indicum CYP81Q1, specifically catalyzes methylenedioxy bridge (MDB) formation in (+)-Epipinoresinol to produce (+)-pluviatilol. Both (+)-Epipinoresinol and (+)-pluviatilol are putative intermediates of (+)-2-episesalatin based on their diastereomeric configurations. On the other hand, CYP81Q3 accepts neither (+)- nor (-)-pinoresinol as a substrate. This diastereomeric selectivity of CYP81Q3 is in clear contrast to that of CYP81Q1, which specifically converts (+)-pinoresinol to (+)-sesamin via (+)-piperitol by the sequential formation of two MDBs but does not accept (+)-Epipinoresinol as a substrate. Moreover, (+)-pinoresinol does not interfere with the conversion of (+)-Epipinoresinol to (+)-pluviatilol by CYP81Q3. Amino acid substitution and CO difference spectral analyses show that polymorphic residues between CYP81Q1 and CYP81Q3 proximal to their putative substrate pockets are crucial for the functional diversity and stability of these two enzymes. Our data provide clues to understanding how the lineage-specific functional differentiation of respective biosynthetic enzymes substantiates the stereoisomeric diversity of lignan structures. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)