| Description: |
Pachypodol has antibacterial and antifungal activities against Bacillus subtilis, Staphylococcus aureus, Staphylococcus faecalis, Echerichia coli, Pseudomonas aeruginosa, Candida albicans, Candida krusei and Candida galabrata. Pachypodol has cytotoxic potential , it can inhibit the growth of CaCo 2 colon cancer cell line in vitro. Pachypodol exhibits anti-emetic effects. |
| In vitro: |
| Phytother Res. 2008 Dec;22(12):1684-7. | | Pachypodol, a flavonol from the leaves of Calycopteris floribunda, inhibits the growth of CaCo 2 colon cancer cell line in vitro.[Pubmed: 18570232] | Calycopteris floribunda Lam., commonly known as 'goichia lata or goache lata', is a large climbing woody shrub from Bangladesh, and well distributed in a number of other south-east Asian countries.
Traditionally, C. floribunda has been used in colic, as an antihelminthic, astringent and carminative, and for the treatment of diarrhoea, dysentery, jaundice and malaria in many countries including Bangladesh.
METHODS AND RESULTS:
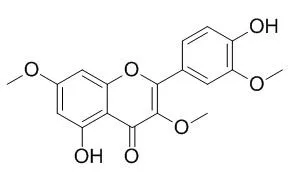
Pachypodol (5,4'-dihydroxy-3,7,3'-trimethoxyflavone) has been isolated from the leaves of C. floribunda by repeated column chromatography on silica gel, and the structure confirmed by spectroscopic means. While the general toxicity of Pachypodol was determined by the brine shrimp lethality assay, the cytotoxic potential of this flavonoid has been evaluated by the Promega's CellTiter 96 Non-Radioactive Cell Proliferation Assay using the CaCo-2 colon cancer cell line (IC(50) = 185.6 microM). A summary of the biological activities of Pachypodol reported to date is also presented. | | J Agric Food Chem. 2006 Feb 22;54(4):1217-21. | | Pachypodol from Croton ciliatoglanduliferus Ort. as water-splitting enzyme inhibitor on thylakoids.[Pubmed: 16478239] |
METHODS AND RESULTS:
A bioactivity-guided chemical study of aerial parts of Croton ciliatoglanduliferus Ort. led to the isolation for the first time of the flavonoids retusin (5-hydroxy-3,7,3',4'-tetramethoxyflavone) (1) and Pachypodol (5,4'-dihydroxy-3,7,3'-trimethoxyflavone) (2) from the n-hexane extract. Compounds 1 and 2 were separated by preparative thin-layer chromatography. Compound 2 was the most active compound on ATP synthesis inhibition. The I50 value was 51 microM. Pachypodol behaves as a Hill reaction inhibitor. It inhibited the uncoupled electron flow on photosystem II partial reaction from water to dichlorophenol indophenol (DCPIP) and from water to sodium silicomolybdate. However, the uncoupled partial reaction from diphenylcarbazide to DCPIP and the uncoupled photosystem I from DCPIPred to MV were not inhibited by 2. These results were corroborated by fluorescence decay data.
CONCLUSIONS:
Therefore, Pachypodol inhibits the water-splitting enzyme activity. Compound 1 with a 4'-methoxy group was a weak inhibitor, indicating that the 4' free -OH group is important for strong inhibition. | | Pharm. Biol., 2003, 41(7):483-6. | | Antifungal Flavonoids from Ballota glandulosissima.[Reference: WebLink] |
METHODS AND RESULTS:
The flavonoids kumatakenin (1), Pachypodol (2), 5-hydroxy-7,3′,4′-trimethoxyflavone (3), velutin (4), salvigenin (5), retusin (6) and corymbosin (7) have been isolated from the aerial parts of Ballota glandulosissima Hub.-Mor & Patzak. Among them, 2–4 and 7 have not been reported previously in the genus Ballota. The antibacterial and antifungal activities of 1–4 and 6 were tested against Bacillus subtilis, Staphylococcus aureus, Staphylococcus faecalis, Echerichia coli, Pseudomonas aeruginosa, Candida albicans, Candida krusei and Candida galabrata.
| | Phytomedicine. 1999 May;6(2):89-93. | | Anti-emetic principles of Pogostemon cablin (Blanco) Benth.[Pubmed: 10374246 ] |
METHODS AND RESULTS:
Bioassay-guided fractionation of anti-emetic extracts and constituents of 8 traditional Chinese herbal drugs was performed. Twenty extracts described in Table 1 showed anti-emetic activity on copper sulfate induced-emesis in young chicks. From the n-hexane extract of Pogostemon cablin, patchouli alcohol (1), pogostol (2), stigmast-4-en-3-one (3), retusin (4), and Pachypodol (5) were tested and exhibited anti-emetic effects. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)