| Description: |
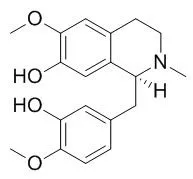
Reticuline is a key compound in the biosynthetic pathway for isoquinoline alkaloids in plants, which include morphine, codeine and berberine. Reticuline possesses potent central nervous system depressant action, it (50-100 mg/kg i.p.) can produce alteration of behaviour pattern, prolongation of pentobarbital-induced sleep, reduction in motor coordination and D-amphetamine-induced hypermotility and suppression of the conditioned avoidance response. (S)-Reticuline can elicit vasorelaxation probably due to the blockade of the L-type voltage-dependent Ca(2+) current in rat aorta, the effect may contribute to the potential cardioprotective efficacy of (S)-reticuline. |
| Targets: |
Calcium Channel |
| In vitro: |
| Transgenic Res. 2007 Jun;16(3):363-75. | | Knockdown of berberine bridge enzyme by RNAi accumulates (S)-reticuline and activates a silent pathway in cultured California poppy cells.[Pubmed: 17103244 ] | Reticuline is a key compound in the biosynthetic pathway for isoquinoline alkaloids in plants, which include morphine, codeine and berberine.
METHODS AND RESULTS:
We established cultured California poppy (Eschscholzia californica) cells, in which berberine bridge enzyme (BBE) was knocked down by RNA interference, to accumulate the important key intermediate Reticuline. Both BBE mRNA accumulation and enzyme activity were effectively suppressed in transgenic cells. In these transgenic cells, end-products of isoquinoline alkaloid biosynthesis, such as sanguinarine, were considerably reduced and Reticuline was accumulated at a maximum level of 310 mug/g-fresh weight. In addition, 1 g-fresh weight of these cells secreted significant amounts of Reticuline into the medium, with a maximum level of 6 mg/20 mL culture medium. These cells also produced a methylated derivative of Reticuline, laudanine, which could scarcely be detected in control cells.
CONCLUSIONS:
We discuss the potential application of RNAi technology in metabolic modification and the flexibility of plant secondary metabolism. | | Med Sci Monit. 2005 May;11(5):MS1-5. | | In vivo and in vitro L-DOPA and reticuline exposure increases ganglionic morphine levels.[Pubmed: 15874894] | Given the presence of morphine, its metabolites and precursors, i.e., Reticuline, in mammalian and invertebrate tissues, it has become imperative to determine if exposing tissues to putative opiate alkaloid and dopamine precursors would result in increasing endogenous morphine levels.
METHODS AND RESULTS:
Endogenous morphine levels were determined by high performance liquid chromatography coupled to electrochemical detection and radioimmunoassay, following incubation of Mytilus edulis pedal ganglia with Reticuline or L-DOPA. Injection of L-DOPA or Reticuline into healthy animals was via the foot.
Ganglia incubated in vitro with Reticuline or L-DOPA for 1 hour exhibited a concentration and time dependent statistically significant increase in their endogenous morphine levels (5.0 +/- 0.47, 3.6 +/- 0.45 ng/ganglion, respectively). Injection of intact, healthy animals with Reticuline or L-DOPA also results in significantly higher endogenous ganglionic morphine levels.
METHODS AND RESULTS:
Taken together, we show that L-DOPA is being converted to morphine, demonstrating that pedal ganglia can synthesize morphine from these putative precursors in vitro and in vivo. This is the first demonstration of morphine being synthesized in a normal, healthy free living animal. |
|
| In vivo: |
| J Ethnopharmacol. 1998 Aug;62(1):57-61. | | Central depressant effects of reticuline extracted from Ocotea duckei in rats and mice.[Pubmed: 9720612] |
METHODS AND RESULTS:
Neuropharmacological studies were carried out with Reticuline, a benzylisoquinoline alkaloid, isolated from Ocotea duckei Vattimo. It was found that Reticuline (50-100 mg/kg i.p.) produced alteration of behaviour pattern, prolongation of pentobarbital-induced sleep, reduction in motor coordination and D-amphetamine-induced hypermotility and suppression of the conditioned avoidance response.
CONCLUSIONS:
These observations suggest that Reticuline possesses potent central nervous system depressant action. | | Planta Med . 2018 Jan;84(1):20-25. | | Anti-Inflammatory Effects of Boldine and Reticuline Isolated from Litsea cubeba through JAK2/STAT3 and NF-κB Signaling Pathways[Pubmed: 28651290] | | Abstract
The anti-inflammatory effects of boldine and Reticuline isolated from Litsea cubeba were evaluated by using xylene-induced ear edema and carrageenan-induced paw edema in mice and rats. Our results demonstrated that intragastric administration with boldine and Reticuline significantly mitigated ear weight in mice and decreased paw volume in rats. A combination administration of boldine (0.5 mg/kg) + Reticuline (0.25 mg/kg) resulted in a potentiated inhibition in these two models. In parallel, boldine or Reticuline reduce the infiltration of neutrophil leukocytes in rat paw tissue, respectively, and the combination of the two groups performed a better anti-inflammatory activity as shown in histopathologies. Boldine, Reticuline, and their combination notably inhibited mRNA expressions of TNF-α, and IL-6 and reduced the phosphorylation levels of Janus kinase 2 (JAK2) and signal transducer and activator of transcription 3 (STAT3). Beyond that, their combination also can reduce the phosphorylation levels of p65 and IκBα in the pathological tissues of animals, as observed by real-time PCR and western blot analyses, respectively. These findings indicate for the first time that boldine and Reticuline have not only anti-inflammatory activity but also potential synergistic effects in vivo. The underlying mechanism may relate to the inhibition on the expression of pro-inflammatory cytokines, such as TNF-α and IL-6, which may be a consequence of JAK2/STAT3 and NF-κB pathway involvements. This study provides useful data for further exploration and application of boldine and Reticuline as potential anti-inflammatory medicines. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)