| In vitro: |
| Phytomedicine. 2003;10(6-7):544-51. | | Study on the inhibitory effects of Korean medicinal plants and their main compounds on the 1,1-diphenyl-2-picrylhydrazyl radical.[Pubmed: 13678241] |
METHODS AND RESULTS:
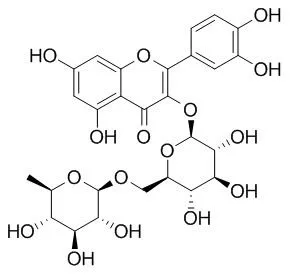
The polyphenols isolated from these plants, procyanidin B-3, (+)-catechin, gallic acid, methyl gallate, quercetin, quercetin-3-O-beta-D-glucoside, quercetin-3-O-beta-galactoside, Quercetin-3-o-rutinose and kaempferol, exerted strong DPPH radical-scavenging activity.
CONCLUSIONS:
These results suggest that the Korean medicinal plants and the polyphenols isolated from them that exhibited effective radical-scavenging activity may be promising agents for scavenging free radicals and treating diseases associated with excess free radicals. | | Am J Chin Med. 2003;31(6):907-17. | | The inhibitory effects of 12 medicinal plants and their component compounds on lipid peroxidation.[Pubmed: 14992543] |
The antioxidative activities of 12 medicinal plants and the compounds isolated from them were investigated using the thiocyanate method to evaluate inhibitory effects on lipid peroxidation in the linoleic acid system.
METHODS AND RESULTS:
The peroxide levels gradually increased during incubation in the presence of linoleic acid over 3 days, and most of the plants inhibited lipid peroxidation. In particular, of the plants tested, Cudrania tricuspidata, Zanthoxylum piperitum, Houttuynia cordata and Ulmus parvifolia reduced lipid peroxidation more effectively as lipid peroxidation progressed, resulting in inhibition of about 80% relative to the control value by the 3rd day of incubation. In addition, the polyphenols isolated from the plants also showed marked and dose-dependent inhibitory effects on lipid peroxidation. The compounds with the strongest activities were 3,4-dihydroxylbenzoic acid, quercetin, the quercetin glycosides quercetin-3-O-beta-D-galactoside, quercetin-3-O-alpha-L-rhamnoside, quercetin-3-O-beta-D-glucoside and Quercetin-3-o-rutinose, catechin, gallic acid, methyl gallate and rosamultin isolated from Zanthoxylum piperitum, Houttuynia cordata, Rosa rugosa and Cedrela sinensis. Moreover, quercetin glycosides showed stronger activity than quercetin, suggesting that glycosylation increases the antioxidative activity of quercetin.
CONCLUSIONS:
Our results indicate that the medicinal plants and their polyphenols show promise as therapeutic agents for various disorders involving free radical reactions. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)