The relationships between inhibition of platelet prostaglandin (PG) synthesis and aggregation, and suppression inflammation were investigated with a number of benzoic acid (aspirin-like) chemicals.
METHODS AND RESULTS:
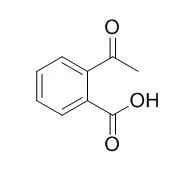
The compounds studied were 2-Acetylbenzoic acid (ABA), 3-methylphthalide (3-MP), 3-propionyloxybenzoic acid (3-PBA) and 2-propionyloxybenzoic acid (2-PBA). At 0.5--0.6 mM, 3-MP inhibited the second phase of ADP-induced aggregation in human platelets, and reduced collagen-induced aggregation by 50%. Previous studies have shown 2-PBA to inhibit aggregation at similar concentrations. In contrast, ABA required 10 times higher concentrations, and low concentrations actually potentiated aggregation. Inhibition of PG synthesis from 14C-arachidonic acid (AA) by human platelets was shown for 2-PBA, but not to 3-BPA, or ABA. At high concentration (1 mM), 3-MP showed modest inhibitory activity. Significant inhibition of AA aggregation was produced by ASA (83%), 2-PBA (76%) and 3-MP (69%), an order reflecting their inhibition of PG synthesis, where ABA and 3-PBA did not inhibit AA aggregation. Carrageenin-induced edema of the rat paw was suppressed by 3-MP, ABA and 2-PBA; all being roughly equipotent with aspirin. In contrast, 3-PBA did not suppress edema. Following oral administration of the drugs to rats, PG synthesis from labeled AA by rat platelets showed similar profiles to effects of the drugs on PG synthesis in human platelets.
CONCLUSIONS:
This suggests that biotransformation or species differences are not explanations for the observed differences in activity in the various test systems. The results indicate that, in a related series of chemicals there is not a good correlation between ability to inhibit platelet PG synthesis, anti-aggregatory activity and anti-inflammatory activity. Multiple mechanisms of action, differing sensitivities of various tissue PG synthetases, or unidentified factors could be involved. |





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)