| In vitro: |
| Plant Physiol., 1989, 91(3):842-7. | | A Chalcone and Two Related Flavonoids Released from Alfalfa Roots Induce nod Genes of Rhizobium meliloti.[Pubmed: 16667146 ] | Flavonoid signals from alfalfa (Medicago sativa L.) induce transcription of nodulation (nod) genes in Rhizobium meliloti. Previous investigations identified the flavone luteolin as an active inducer in alfalfa seed extracts, but the nature of nod inducers released from roots has not been reported.
METHODS AND RESULTS:
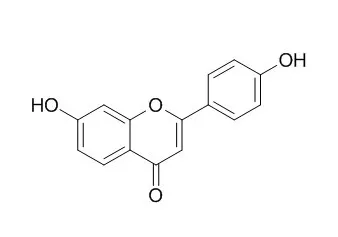
Root exudate from 3-day-old alfalfa seedlings was purified and then assayed for biological activity with a nodABC-lacZ fusion in R. meliloti. Indentities of major nod inducers were established by spectroscopic analyses (ultraviolet/visible, proton nuclear magnetic resonance, and mass spectroscopy) and comparison with authentic standards. Major nod inducers, which were identified as 4',7-Dihydroxyflavone, 4'-7-dihydroxyflavanone, and 4,4'-dihydroxy-2'-methoxychalcone, were released from seedling roots at 54, 22, and 20 picomole.plant(-1).day(-1), respectively. Luteolin was not found in these root exudates.
CONCLUSIONS:
The 4,4'-dihydroxy-2'-methoxychalcone induced nod genes at a concentration one order of magnitude lower than luteolin and is the first naturally released chalcone reported to have this function. Moderate and weak nod-inducing activity was associated, respectively, with 4',7-Dihydroxyflavone and 4',7-dihydroxyflavanone. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)