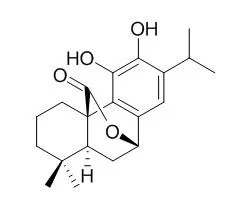
| Description: |
Carnosol, a novel agonist of TRPA1 with an EC50 value of 12.46 uM, which exhibits anti-inflammatory, anti-nociceptive, hepatoprotective, antioxidant, anticarcinogen, anti-angiogenic, anti- invasive and antimetastatic properties. Carnosol can cause a significant decrease in both bacterial and yeast growth whilst, it may prove useful as a food antioxidant which could also contribute to the retardation of the microbial spoilage of foods; it also can inhibit adipocyte differentiation in mouse 3T3-L1 cells through induction of phase2 enzymes and activation of glutathione metabolism, it may be a potential drug against obesity-related diseases. |
| In vitro: |
| Eur J Nutr. 2013 Feb;52(1):85-95. | | Anti-angiogenic properties of carnosol and carnosic acid, two major dietary compounds from rosemary.[Pubmed: 22173778 ] | The use of rosemary (Rosmarinus officinalis) leaves and their constituents as a source of dietary antioxidants and flavoring agents is continuously growing. Carnosol and carnosic acid, two major components of rosemary extracts, have shown activity for cancer prevention and therapy.
In this study, we investigate the cytotoxic and anti-angiogenic activities of Carnosol and carnosic acid, in order to get further insight into their mechanism of action.
METHODS AND RESULTS:
Our results demonstrate that the mentioned diterpenes inhibit certain functions of endothelial cells, namely, differentiation, proliferation, migration and proteolytic capability. Our data indicate that their growth inhibitory effect, exerted on proliferative endothelial and tumor cells, could be due to, at least in part, an induction of apoptosis. Inhibition of the mentioned essential steps of in vitro angiogenesis agrees with the observed inhibition of the in vivo angiogenesis, substantiated by using the chick chorioallantoic membrane assay.
CONCLUSIONS:
The anti-angiogenic activity of Carnosol and carnosic acid could contribute to the chemopreventive, antitumoral and antimetastatic activities of rosemary extracts and suggests their potential in the treatment of other angiogenesis-related malignancies. | | Food Microbiol., 1987, 4(4):311-5. | | Antimicrobial activity of carnosol and ursolic acid: two anti-oxidant constituents of Rosmarinus officinalis L.[Reference: WebLink] |
METHODS AND RESULTS:
Inhibition of the growth of 6 strains of food associated bacteria and yeasts by Carnosol and Ursolic acid, two antioxidant compounds extracted from rosemary, was investigated and compared to inhibitory effects exhibited by the commonly used food antioxidants Butylated Hydroxyanisole (BHA) and Butylated Hydroxytoluene (BHT). At the highest concentration used (150 μg ml−1) Carnosol inhibited all the test microbes to the greatest extent. BHA proved a superior inhibitor to Ursolic acid which itself was more effective overall than BHT. Even at 50 μg ml−1, Carnosol caused a significant decrease in both bacterial and yeast growth whilst BHA proved more effective against the yeasts than bacteria.
CONCLUSIONS:
Thus Carnosol might prove useful as a food antioxidant which could also contribute to the retardation of the microbial spoilage of foods. | | Oncotarget . 2018 Feb 6;9(76):34200-34212. | | Carnosol suppresses patient-derived gastric tumor growth by targeting RSK2[Pubmed: 30344937] | | Abstract
Carnosol is a phenolic diterpene that is isolated from rosemary, sage, and oregano. It has been reported to possess anti-oxidant, anti-inflammatory, and anti-cancer properties. However, the molecular mechanism of Carnosol's activity against gastric cancer has not been investigated. Herein, we report that Carnosol is an RSK2 inhibitor that attenuates gastric cancer growth. Carnosol reduced anchorage-dependent and -independent gastric cancer growth by inhibiting the RSKs-CREB signaling pathway. The results of in vitro screening and cell-based assays indicated that Carnosol represses RSK2 activity and its downstream signaling. Carnosol increased the G2/M phase and decreased S phase cell cycle and also induced apoptosis through the activation of caspases 9 and 7 and inhibition of Bcl-xL expression. Notably, oral administration of Carnosol suppressed patient-derived gastric tumor growth in an in vivo mouse model. Our findings suggest that Carnosol is an RSK2 inhibitor that could be useful for treating gastric cancer.
Keywords: RSK2; Carnosol; gastric cancer; patient-derived tumor xenograft. | | Int J Mol Sci . 2019 Feb 18;20(4):880. | | Carnosol as a Nrf2 Activator Improves Endothelial Barrier Function Through Antioxidative Mechanisms[Pubmed: 30781644] | | Abstract
Oxidative stress is the main pathogenesis of diabetic microangiopathy, which can cause microvascular endothelial cell damage and destroy vascular barrier. In this study, it is found that Carnosol protects human microvascular endothelial cells (HMVEC) through antioxidative mechanisms. First, we measured the antioxidant activity of Carnosol. We showed that Carnosol pretreatment suppressed tert-butyl hydroperoxide (t-BHP)-induced cell viability, affected the production of lactate dehydrogenase (LDH) as well as reactive oxygen species (ROS), and increased the produce of nitric oxide (NO). Additionally, Carnosol promotes the protein expression of vascular endothelial cadherin (VE-cadherin) to keep the integrity of intercellular junctions, which indicated that it protected microvascular barrier in oxidative stress. Meanwhile, we investigated that Carnosol can interrupt Nrf2-Keap1 protein-protein interaction and stimulated antioxidant-responsive element (ARE)-driven luciferase activity in vitro. Mechanistically, we showed that Carnosol promotes the expression of heme oxygenase 1(HO-1) and nuclear factor-erythroid 2 related factor 2(Nrf2). It can also promote the expression of endothelial nitric oxide synthase (eNOS). Collectively, our data support the notion that Carnosol is a protective agent in HMVECs and has the potential for therapeutic use in the treatments of microvascular endothelial cell injury.
Keywords: HMVEC cells; antioxidant activity; Carnosol; microvascular endothelial protection. |
|
| In vivo: |
| Eur J Gastroenterol Hepatol. 2002 Sep;14(9):1001-6. | | Protective effect of carnosol on CCl(4)-induced acute liver damage in rats.[Pubmed: 12352220] | We recently reported that (Lamiaceae) may alleviate CCl(4)-induced acute hepatotoxicity in rats, possibly blocking the formation of free radicals generated during CCl(4) metabolism. Carnosol, one of the main constituents of Rosmarinus, has been shown to have antioxidant and scavenging activities. Therefore, it is plausible to expect that Carnosol may mediate some of the effects of Rosmarinus on oxidative stress consequences induced by CCl(4) in the liver.
We evaluated the effectiveness of Carnosol to normalize biochemical and histological parameters of CCl(4)-induced acute liver injury.
METHODS AND RESULTS:
Male Sprague Dawley rats (n = 5) injured by CCl(4) (oral dose 4 g/kg of body weight) were treated with a single intraperitoneal dose (5 mg/kg) of Carnosol. Twenty-four hours later, the rats were anaesthetized deeply to obtain the liver and blood, and biochemical and histological parameters of liver injury were evaluated.
Carnosol normalized bilirubin plasma levels, reduced malondialdehyde (MDA) content in the liver by 69%, reduced alanine aminotransferase (ALT) activity in plasma by 50%, and partially prevented the fall of liver glycogen content and distortion of the liver parenchyma.
CONCLUSIONS:
Carnosol prevents acute liver damage, possibly by improving the structural integrity of the hepatocytes. To achieve this, Carnosol could scavenge free radicals induced by CCl(4), consequently avoiding the propagation of lipid peroxides. It is suggested that at least some of the beneficial properties of Rosmarinus officinalis are due to Carnosol. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)