| In vitro: |
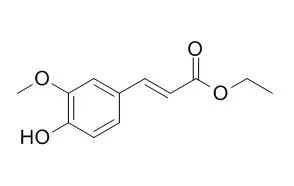
| Molecules. 2014 Jun 17;19(6):8124-39. | | Ethyl ferulate, a component with anti-inflammatory properties for emulsion-based creams.[Pubmed: 24941338] | Ethyl ferulate (FAEE) has been widely studied due to its beneficial heath properties and, when incorporated in creams, shows a high sun protection capacity.
METHODS AND RESULTS:
Here we aimed to compare Ethyl ferulate and its precursor, ferulic acid (FA), as free radical scavengers, inhibitors of oxidants produced by leukocytes and the alterations in rheological properties when incorporated in emulsion based creams. The cell-free antiradical capacity of Ethyl ferulate was decreased compared to FA. However, Ethyl ferulate was more effective regarding the scavenging of reactive oxygen species produced by activated leukocytes. Stress and frequency sweep tests showed that the formulations are more elastic than viscous. The viscoelastic features of the formulations were confirmed in the creep and recovery assay and showed that the Ethyl ferulate formulation was less susceptive to deformation. Liberation experiments showed that the rate of Ethyl ferulate release from the emulsion was slower compared to FA.
CONCLUSIONS:
In conclusion,Ethyl ferulate is more effective than FA as a potential inhibitor of oxidative damage produced by oxidants generated by leukocytes. The rheological alterations caused by the addition of Ethyl ferulate are indicative of lower spreadability, which could be useful for formulations used in restricted areas of the skin. | | Journal of the Fourth Military Medical University, 2002,23(6):537-9. | | Inhibitive effect and mechanism of Ethyl ferulate on platelet congregation induced by ADP.[Reference: WebLink] | To investigate the inhibitory actions of Ethyl ferulate on platelet congregate induced by ADP, and the effect of platelet intracellular calcium oscillations.
METHODS AND RESULTS:
To observe platelet congregate rate induced by congregater TYXN-91. 200 mL·L~(-1) polyethylene glycol 400 as contral. Platelet intracellular calcium oscillation were observed by laser scanning Ethyl ferulate Inhibitions rate were (%) 26.3 ± 3.3, 33.4 ± 2.4, 73.4 ± 3.1 and 94.9 ± 2.7, (n = 8) at different concentration (0.1, 0.5, 1.5 and 3.0 mmol·L~(-1)), significantly increased than that of the group of ferulic acid, the change of ΔFI 4.6 ± 1.7 is much lower than resting level 10.3 ± 2.6(n = 8, P < 0.01).
CONCLUSIONS:
The inhibitive effect of Ethyl ferulate on platelet congregation induced by ADP was much more than that of ferulic acid.
|
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)