| In vitro: |
| Biol. Pharm. Bull., 2008, 31(5):908-15. | | Re-evaluation of the antioxidant prenylated flavonoids from the roots of Sophora flavescens.[Pubmed: 18451517] |
METHODS AND RESULTS:
The objective of this research was to re-evaluate the antioxidant effects of the prenylated flavonoids from Sophora flavescens via in vitro 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2'-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS), peroxynitrite (ONOO(-)), and total reactive oxygen species (ROS) assays. In addition, a further examination of kuraridinol, kurarinol, and kurarinone, also isolated from S. flavescens, was carried out by the inhibition of tert-butylhydroperoxide (t-BHP)-induced intracellular ROS generation and t-BHP-induced activation of nuclear factor-kappaB (NF-kappaB). Upon re-examination of the ethyl acetate (EtOAc) soluble fraction of S. flavescens, two major prenylated chalcones, including kuraridin and kuraridinol, along with a minor prenylated flavonol, Kushenol C, were isolated as good DPPH scavengers. This was in contrast to the prenylated flavanones, sophoraflavanone G and kurarinone, which were isolated from the methylene chloride (CH(2)Cl(2)) fraction of the same source. Five flavanones consisting of kushenol E, leachianone G, kurarinol, sophoraflavanone G, and kurarinone exhibited significant antioxidant potentials in the ABTS, ONOO(-), and total ROS assays; however, the prenylated chalcones and prenylated flavonol showed more potent scavenging/inhibitory activities than the prenylated flavanones. Therefore, the prenylated chalcones and prenylated flavonol, rather than the prenylated flavanones, may make important contributions toward the marked antioxidant capacities of S. flavescens. Furthermore, kuraridinol, kurarinol, and kurarinone showed significant inhibitory activities against intracellular ROS levels as well as NF-kappaB activation by t-BHP.
CONCLUSIONS:
Overall, the results indicate that S. flavescens and its prenylated flavonoids may possess good anti-inflammatory activity, which is implicated in their significant antioxidant activity. | | Molecules . 2020 Apr 12;25(8):1768. | | In vitro Anti-Inflammatory and Anti-Oxidative Stress Activities of Kushenol C Isolated from the Roots of Sophora flavescens[Pubmed: 32290603] | | Abstract
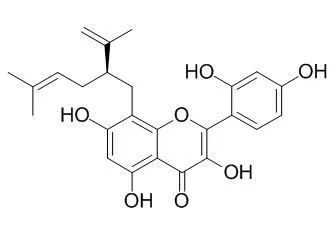
Kushenol C (KC) is a prenylated flavonoid isolated from the roots of Sophora flavescens aiton. Little is known about its anti-inflammatory and anti-oxidative stress activities. Here, we investigated the anti-inflammatory and anti-oxidative stress effects of KC in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages, and tert-butyl hydroperoxide (tBHP)-induced oxidative stress in HaCaT cells. The results demonstrated that KC dose-dependently suppressed the production of inflammatory mediators, including NO, PGE2, IL-6, IL1β, MCP-1, and IFN-β in LPS-stimulated RAW264.7 macrophages. The study demonstrated that the inhibition of STAT1, STAT6, and NF-κB activations by KC might have been responsible for the inhibition of NO, PGE2, IL-6, IL1β, MCP-1, and IFN-β in the LPS-stimulated RAW264.7 macrophages. KC also upregulated the expression of HO-1 and its activities in the LPS-stimulated RAW264.7 macrophages. The upregulation of Nrf2 transcription activities by KC in the LPS-stimulated RAW264.7 macrophages was demonstrated to be responsible for the upregulation of HO-1 expression and its activity in LPS-stimulated RAW264.7 macrophages. In HaCaT cells, KC prevented DNA damage and cell death by upregulating the endogenous antioxidant defense system involving glutathione, superoxide dismutase, and catalase, which prevented reactive oxygen species production from tert-butyl hydroperoxide (tBHP)-induced oxidative stress in HaCaT cells. The upregulated activation of Nrf2 and Akt in the PI3K-Akt signaling pathway by KC was demonstrated to be responsible for the anti-oxidative stress activity of KC in HaCaT cells. Collectively, the study suggests that KC can be further investigated as a potential anti-inflammatory candidate for the treatment of inflammatory diseases.
Keywords: Sophora flavescens; anti-inflammation; anti-oxidative stress; Kushenol C. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)