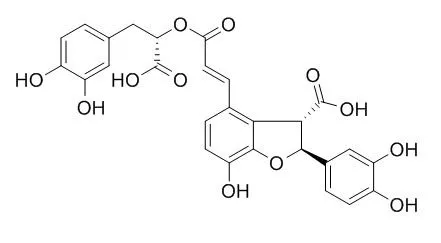
| Description: |
Lithospermic acid has anti-HIV, antioxidant ,anti-inflammatory, and hepatoprotective effects, is a competitive inhibitor of xanthine oxidas (XO), can directly scavenge superoxide and inhibit superoxide production in vitro, and presents hypouricemic actions in vivo. Lithospermic acid has inhibitory effects on proliferation and migration of rat vascular smooth muscle cells, it has a preventive effect on the development of diabetic retinopathy. Lithospermic acid can attenuate 1-methyl-4-phenylpyridine-induced neurotoxicity by blocking neuronal apoptotic and neuroinflammatory pathways. Lithospermic acid can attenuate mesenteric ischemia reperfusion injury in rat intestines by increasing tissue SOD and GPx activities and decreasing MDA and MPO levels, also improves morphological alterations which occurred after periods of reperfusion. |
| In vivo: |
| Adv Clin Exp Med. 2012 Jul-Aug;21(4):433-9. | | Lithospermic acid and ischemia/reperfusion injury of the rat small intestine prevention.[Pubmed: 23240448] | Intestinal ischemia and reperfusion (I-R) injury of different causes, including cardiac insufficiency, sepsis, vasodepressant and cardiodepressant drugs, and complications of long-lasting surgery, represents a major clinical problem.
The purpose of the present study was to investigate whether Lithospermic acid (LA) can reduce oxidative stress and histological damage in the rat small bowel subjected to mesenteric I-R injury.
METHODS AND RESULTS:
The study was performed on three groups of animals, each composed of 7 rats: the SO (sham operation) group, the I-R/Untreated group and the I-R/LA (I-R plus LA pretreatment) group. Intestinal ischemia for 45 minutes and reperfusion for 60 minutes were applied. Ileum specimens were obtained to determine the tissue level of malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GPx), catalase (CAT) and myeloperoxidase (MPO) activities and histological changes.
Untreated intestinal I-R resulted in increased tissue MDA and MPO levels and diminished SOD and GPx activities. These changes were found to be almost reversed in the LA treatment group. Histopathologically, the intestinal injury in rats treated with LA was less than the untreated I-R group.
CONCLUSIONS:
Lithospermic acid attenuates mesenteric ischemia reperfusion injury in rat intestines by increasing tissue SOD and GPx activities and decreasing MDA and MPO levels. Lithospermic acid also improves morphological alterations which occurred after periods of reperfusion. | | PLoS One. 2014 Jun 6;9(6):e98232. | | The effect of lithospermic acid, an antioxidant, on development of diabetic retinopathy in spontaneously obese diabetic rats.[Pubmed: 24905410] | Lithospermic acid B (LAB), an active component isolated from Salvia miltiorrhiza radix, has been reported to have antioxidant effects. We examined the effects of LAB on the prevention of diabetic retinopathy in Otsuka Long-Evans Tokushima Fatty (OLETF) rats, an animal model of type 2 diabetes.
METHODS AND RESULTS:
LAB (10 or 20 mg/kg) or normal saline were given orally once daily to 24-week-old male OLETF rats for 52 weeks. At the end of treatment, fundoscopic findings, vascular endothelial growth factor (VEGF) expression in the eyeball, VEGF levels in the ocular fluid, and any structural abnormalities in the retina were assessed. Glucose metabolism, serum levels of high-sensitivity C-reactive protein (hsCRP), monocyte chemotactic protein-1 (MCP1), and tumor necrosis factor-alpha (TNFα) and urinary 8-hydroxy-2'-deoxyguanosine (8-OHdG) levels were also measured. Treatment with LAB prevented vascular leakage and basement membrane thickening in retinal capillaries in a dose-dependent manner. Insulin resistance and glucose intolerance were significantly improved by LAB treatment. The levels of serum hsCRP, MCP1, TNFα, and urinary 8-OHdG were lower in the LAB-treated OLETF rats than in the controls.
CONCLUSIONS:
Treatment with LAB had a preventive effect on the development of diabetic retinopathy in this animal model, probably because of its antioxidative effects and anti-inflammatory effects. | | Chem Biol Interact. 2008 Nov 25;176(2-3):137-42. | | Lithospermic acid as a novel xanthine oxidase inhibitor has anti-inflammatory and hypouricemic effects in rats.[Pubmed: 18694741 ] | Lithospermic acid (LSA) was originally isolated from the roots of Salvia mitiorrhiza, a common herb of oriental medicine. Previous studies demonstrated that LSA has antioxidant effects. In this study, we investigated the in vitro xanthine oxidase (XO) inhibitory activity, and in vivo hypouricemic and anti-inflammatory effects of rats.
METHODS AND RESULTS:
XO activity was detected by measuring the formation of uric acid or superoxide radicals in the xanthine/xanthine oxidase system. The results showed that LSA inhibited the formation of uric acid and superoxide radicals significantly with an IC50 5.2 and 1.08 microg/ml, respectively, and exhibited competitive inhibition. It was also found that LSA scavenged superoxide radicals directly in the system beta-NADH/PMS and inhibited the production of superoxide in human neutrophils stimulated by PMA and fMLP. LSA was also found to have hypouricemic activity on oxonate-pretreated rats in vivo and have anti-inflammatory effects in a model of gouty arthritis.
CONCLUSIONS:
These results suggested that LSA is a competitive inhibitor of XO, able to directly scavenge superoxide and inhibit superoxide production in vitro, and presents hypouricemic and anti-inflammatory actions in vivo. | | Oncol Rep . 2015 Aug;34(2):673-80. | | Anti-oxidative and hepatoprotective effects of lithospermic acid against carbon tetrachloride-induced liver oxidative damage in vitro and in vivo[Pubmed: 26081670] | | Abstract
Accumulation of an excess amount of reactive oxygen species (ROS) can cause hepatotoxicity that may result in liver damage. Therefore, development of anti-oxidative agents is needed for reducing liver toxicity. This study investigated the anti-oxidative and hepatoprotective activity of Lithospermic acid, a plant-derived polycyclic phenolic carboxylic acid isolated from Salvia miltiorrhiza, on carbon tetrachloride (CCl4)-induced acute liver damage in vitro and in vivo. The results of the DPPH assay indicated that Lithospermic acid was a good anti-oxidant. the CCl4-exposed Huh7 cell line exhibited decreased cell viability, increased necrosis and elevated ROS and caspase-3/7 activity. Lithospermic acid significantly attenuated the CCl4-induced oxidative damage in a concentration-dependent manner. The result of an in vivo study with BALB/c mice corresponded with the anti-oxidative activity noted in the in vitro study. Exposure of mice to CCl4 resulted in a greater than 2-fold elevation in serum aspartate transaminase (AST) and alanine transaminase (ALT). levels In addition, CCl4-intoxication led to an over 20% decrease in the level of intracellular hepatic enzymes including superoxide dismutase (SOD) and catalase (CAT) as well as increased lipid peroxidation. Upon histological examination of the CCl4-exposed mice, the mouse livers showed severe hepatic damage with a huge section of necrosis and structural destruction. Pretreatment of mice with Lithospermic acid for six days significantly reduced CCl4-induced hepatic oxidative damage, serum AST and ALT. The pretreatment also increased SOD and CAT. The findings suggest that the health status of the liver was improved comparable to the control group after a high-dose treatment with Lithospermic acid (100 mg/kg weight). The potential applicability of Lithospermic acid as a hepatoprotective agent was demonstrated. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)