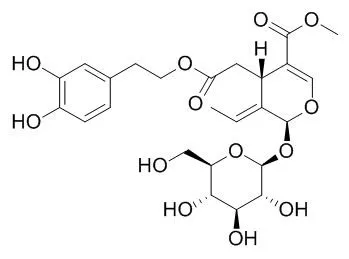
| Description: |
Oleuropein exhibits anti-ischemic, antioxidative, hypolipidemic, in vitro antimycoplasmal , anti-inflammatory, and anti-cancer effects., it also may be helpful in the prevention of diabetic complications associated with oxidative stress. Oleuropein prevents oxidative myocardial injury induced by ischemia and reperfusion, and reduces viremia in duck hepatitis B virus (DHBV)-infected ducks. Oleuropein reduces TLR and MAPK signaling. |
| In vitro: |
| Mol Med Rep. 2015 Jun;11(6):4617-24. | | Oleuropein induces apoptosis via activation of caspases and suppression of phosphatidylinositol 3-kinase/protein kinase B pathway in HepG2 human hepatoma cell line.[Pubmed: 25634350] | Oleuropein is a polyphenol, that is found in extra‑virgin olive oil. Previous studies have shown that Oleuropein inhibits cell proliferation and induces apoptosis in breast cancer, colorectal cancer and thyroid cancer. The aim of the present study was to investigate the effects of Oleuropein in hepatocellular carcinoma (HCC) cells.
METHODS AND RESULTS:
The results of Cell Counting Kit 8 and flow cytometric analysis indicated that Oleuropein effectively inhibited cell viability and induced apoptosis in HepG2 human hepatoma cells in a dose‑dependent manner, through activation of the caspase pathway. Proapoptotic Bcl‑2 family members, BAX and Bcl‑2, were involved in Oleuropein‑induced apoptosis. The phosphatidylinositol 3‑kinase/protein kinase B (PI3K/AKT) signaling pathway was also shown to be involved in this process. Oleuropein was demonstrated to suppress the expression of activated AKT. In addition, AKT overexpression promoted cell survival following treatment with Oleuropein, while inhibition of AKT promoted cell death. Furthermore, the data demonstrated that Oleuropein induces the production of reactive oxygen species (ROS) and that the function of Oleuropein is, at least partially, ROS‑dependent.
CONCLUSIONS:
These results suggest that Oleuropein may be a promising novel chemotherapeutic agent in hepatocellular carcinoma. | | Int J Antimicrob Agents. 2002 Oct;20(4):293-6. | | In vitro antimycoplasmal activity of oleuropein.[Pubmed: 12385687] |
METHODS AND RESULTS:
The activity of Oleuropein, a phenolic glycoside contained in olive oil, was investigated in vitro against Mycoplasma hominis, Mycoplasma fermentans, Mycoplasma pneumoniae and Mycoplasma pirum. Oleuropein inhibited mycoplasmas at concentrations from 20 to 320 mg/l.
CONCLUSIONS:
The MICs of Oleuropein to M. pneumoniae, M. pirum, M. hominis and M. fermentans were 160, 320, 20 and 20 mg/l, respectively. | | Int J Mol Sci . 2018 Feb 28;19(3):686. | | Potential Health Benefits of Olive Oil and Plant Polyphenols[Pubmed: 29495598] | | Abstract
Beneficial effects of natural plant polyphenols on the human body have been evaluated in a number of scientific research projects. Bioactive polyphenols are natural compounds of various chemical structures. Their sources are mostly fruits, vegetables, nuts and seeds, roots, bark, leaves of different plants, herbs, whole grain products, processed foods (dark chocolate), as well as tea, coffee, and red wine. Polyphenols are believed to reduce morbidity and/or slow down the development of cardiovascular and neurodegenerative diseases as well as cancer. Biological activity of polyphenols is strongly related to their antioxidant properties. They tend to reduce the pool of reactive oxygen species as well as to neutralize potentially carcinogenic metabolites. A broad spectrum of health-promoting properties of plant polyphenols comprises antioxidant, anti-inflammatory, anti-allergic, anti-atherogenic, anti-thrombotic, and anti-mutagenic effects. Scientific studies present the ability of polyphenols to modulate the human immune system by affecting the proliferation of white blood cells, and also the production of cytokines or other factors that participate in the immunological defense. The aim of the review is to focus on polyphenols of olive oil in context of their biological activities.
Keywords: Olea europea; anticancer therapy; hydroxytyrosol; Oleuropein; olive oil; polyphenols. |
|
| In vivo: |
| J Nutr Biochem. 2004 Aug;15(8):461-6. | | Oleuropein prevents oxidative myocardial injury induced by ischemia and reperfusion.[Pubmed: 15302080 ] | The potential protective effects of Oleuropein, a dietary antioxidant of olive oil, has been investigated in the isolated rat heart.
METHODS AND RESULTS:
The organs were subjected to 30 minutes of no-flow global ischemia and then reperfused. At different time intervals, the coronary effluent was collected and assayed for creatine kinase activity as well as for reduced and oxidized glutathione. In addition, the extent of lipid peroxidation was evaluated by measuring thiobarbituric acid reactive substance concentration in cardiac muscle. Pretreatment with 20 microg/g Oleuropein before ischemia resulted in a significant decrease in creatine kinase and reduced glutathione release in the perfusate. The protective effect of Oleuropein against the post-ischemic oxidative burst was investigated by measuring the release, in the coronary effluent, of oxidized glutathione, a sensitive marker of heart's exposure to oxidative stress. Reflow in ischemic hearts was accompanied by a prompt release of oxidized glutathione; in ischemic hearts pretreated with Oleuropein, this release was significantly reduced. Membrane lipid peroxidation was also prevented by Oleuropein. The reported data provide the first experimental evidence of a direct cardioprotective effect of Oleuropein in the acute events that follow coronary occlusion, likely because of its antioxidant properties.
CONCLUSIONS:
This finding strengthens the hypothesis that the nutritional benefit of olive oil in the prevention of coronary heart disease can be also related to the high content of Oleuropein and its derivatives. Moreover, our data, together with the well documented antithrombotic and antiatherogenic activity of olive oil polyphenols, indicate these antioxidants as possible therapeutic tools for the pharmacological treatment of coronary heart disease as well as in the case of cardiac surgery, including transplantation. | | J Ethnopharmacol. 2009 Sep 7;125(2):265-8. | | Antiviral efficacy against hepatitis B virus replication of oleuropein isolated from Jasminum officinale L. var. grandiflorum.[Pubmed: 19580857 ] | Jasminum officinale L. var. grandiflorum (JOG) is a folk medicine used for the treatment of hepatitis in south of China. Phytochemical studies showed that secoiridoid glycosides are the typical constituents of this plant.
The present study was undertaken to evaluate the effect of Oleuropein (Ole) derived from the flowers of JOG on hepatitis B virus (HBV) replication in HepG2 2.2.15 cell line in vitro and duck hepatitis B virus (DHBV) replication in ducklings in vivo.
METHODS AND RESULTS:
The extracellular hepatitis B e antigen (HBeAg) and hepatitis B surface antigen (HBsAg) concentrations in cell culture medium were determined by ELISA. DHBV in duck serum was analyzed by dot blot.
Ole blocks effectively HBsAg secretion in HepG2 2.2.15 cells in a dose-dependent manner (IC(50)=23.2 microg/ml). Ole (80 mg/kg, intraperitoneally, twice daily) also reduced viremia in DHBV-infected ducks.
CONCLUSIONS:
Ole therefore warrants further investigation as a potential therapeutic agent for HBV infection. |
|





 Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019)
Cell. 2018 Jan 11;172(1-2):249-261.e12. doi: 10.1016/j.cell.2017.12.019.IF=36.216(2019) Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019)
Cell Metab. 2020 Mar 3;31(3):534-548.e5. doi: 10.1016/j.cmet.2020.01.002.IF=22.415(2019) Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)
Mol Cell. 2017 Nov 16;68(4):673-685.e6. doi: 10.1016/j.molcel.2017.10.022.IF=14.548(2019)